
Q3C — Tables and List
Guidance for Industry
U.S. Department of Health and Human Services
Food and Drug Administration
Center for Drug Evaluation and Research (CDER)
Center for Biologics Evaluation and Research (CBER)
[June 2017]
ICH
Revision 3

Q3C — Tables and List
Guidance for Industry
Additional copies are available from:
Office of Communications,
Division of Drug Information
Center for Drug Evaluation and Research
Food and Drug Administration
10001 New Hampshire Ave., Hillandale Bldg., 4
th
Floor
Silver Spring, MD 20993-0002
Phone: 855-543-3784 or 301-796-3400; Fax: 301-431-6353
druginfo@fda.hhs.gov
http://www.fda.gov/Drugs/GuidanceComplianceRegulatoryInformation/Guidances/default.htm
or
Office of Communication, Outreach and Development
Center for Biologics Evaluation and Research
Food and Drug Administration
10903 New Hampshire Ave., Bldg. 71, Room 3128
Silver Spring, MD 20993-0002
Phone: 800-835-4709 or 240-402-8010
Email: ocod@fda.hhs.gov
https://www.fda.gov/BiologicsBloodVaccines/GuidanceComplianceRegulatoryInformation/Guidances/default.htm
U.S. Department of Health and Human Services
Food and Drug Administration
Center for Drug Evaluation and Research (CDER)
Center for Biologics Evaluation and Research (CBER)
[June 2017]
ICH
Revision 3

Contains Nonbinding Recommendations
Q3C — Tables and List
Guidance for Industry
1
This guidance represents the current thinking of the Food and Drug Administration (FDA or Agency) on
this topic. It does not establish any rights for any person and is not binding on FDA or the public. You
can use an alternative approach if it satisfies the requirements of the applicable statutes and regulations.
To discuss an alternative approach, contact the FDA office responsible for this guidance as listed on the
title page.
I. INTRODUCTION
This is the companion document for the International Council for Harmonisation of Technical
Requirements for Pharmaceuticals for Human Use (ICH) guidance for industry Q3C Impurities:
Residual Solvents, which makes recommendations as to what amounts of residual solvents are
considered safe in pharmaceuticals.
This document may be updated if proposals for change are submitted to the ICH Secretariat for
consideration by the ICH Q3C Expert Working Group (EWG). If the EWG supports the
proposal for change, the proposal will be submitted to the ICH Assembly for endorsement. Any
proposals that are endorsed by the ICH Assembly will be announced through a notice in the
Federal Register prior to the updating of this document. The guidance was revised in November
2003 to reflect updated recommendations for N-Methylpyrrolidone and Tetrahydrofuran, in
February 2012 to reflect an updated recommendation for cumene, and in October 2016 to reflect
updated recommendations for Triethylamine and Methylisobutylketone.
In general, FDA’s guidance documents do not establish legally enforceable responsibilities.
Instead, guidances describe the Agency's current thinking on a topic and should be viewed only
as recommendations, unless specific regulatory or statutory requirements are cited. The use of
the word should in Agency guidances means that something is suggested or recommended, but
not required.
1
This document was developed within the Expert Working Group (Quality) of the International Council for
Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) and has been subject to
consultation by the regulatory parties, in accordance with the ICH process. This document was endorsed by the ICH
Steering Committee at Step 4 of the ICH process in July 1997. At Step 4 of the process, the final draft is
recommended for adoption to the regulatory agencies. This guidance was published in the Federal Register on
December 24, 1997 (62 FR 67377), and is applicable to drug and biological products.
2
The information included for Methylisobutylketone reflects that included in the Revision of PDE Information for
Methylisobutylketone, which reached Step 4 in November 2016 and was subsequently incorporated into the core
guidance.
1

Contains Nonbinding Recommendations
II. LIST OF SOLVENTS INCLUDED IN THE Q3C GUIDANCE
Solvent Other Names Structure Class
Acetic acid
Ethanoic acid
CH
3
COOH
Class 3
Acetone 2-Propanone CH
3
COCH
3
Propan-2-one
Class 3
Acetonitrile
CH
3
CN
Class 2
Anisole Methoxybenzene
Class 3
Benzene Benzol
Class 1
1-Butanol n-Butyl alcohol CH
3
(CH
2
)
3
OH Class 3
Butan-1-ol
2-Butanol CH
3
CH
2
CH(OH)CH
3
sec-Butyl alcohol
Class 3
Butan-2-ol
Butyl acetate
Acetic acid butyl ester
CH
3
COO(CH
2
)
3
CH
3
Class 3
tert-Butylmethyl ether
2-Methoxy-2-methyl-propane
(CH
3
)
3
COCH
3
Class 3
Carbon tetrachloride
Tetrachloromethane
CCl
4
Class 1
Chlorobenzene
Class 2
Chloroform
Trichloromethane
CHCl
3
Class 2
Cumene Isopropylbenzene
C
6
H
5
-CH(CH
3
)
2
(1-Methyl)ethylbenzene
Class 2
Cyclohexane Hexamethylene Class 2
1,2-Dichloroethane CH
2
ClCH
2
Cl
sym-Dichloroethane
Class 1
Ethylene dichloride
Ethylene chloride
1,1-Dichloroethene 1,1-Dichloroethylene H
2
C=CCl
2
Vinylidene chloride
Class 1
1,2-Dichloroethene 1,2-Dichloroethylene ClHC=CHCl
Acetylene dichloride
Class 2
2

Contains Nonbinding Recommendations
Dichloromethane
Methylene chloride
CH
2
Cl
2
Class 2
1,2-Dimethoxyethane
Ethyleneglycol dimethyl ether
Monoglyme
Dimethyl Cellosolve
H
3
COCH
2
CH
2
OCH
3
Class 2
N,N-
Dimethylacetamide
DMA
CH
3
CON(CH
3
)
2
Class 2
N,N- Dimethylformamide
DMF
HCON(CH
3
)
2
Class 2
Dimethyl sulfoxide
Methylsulfinylmethane
Methyl sulfoxide
DMSO
(CH
3
)
2
SO
Class 3
1,4-Dioxane
p-Dioxane
[1,4]Dioxane
Class 2
Ethanol
Ethyl alcohol
CH
3
CH
2
OH
Class 3
2-Ethoxyethanol
Cellosolve
CH
3
CH
2
OCH
2
CH
2
OH
Class 2
Ethyl acetate
Acetic acid ethyl ester
CH
3
COOCH
2
CH
3
Class 3
Ethyleneglycol
1,2-Dihydroxyethane
1,2-Ethanediol
HOCH
2
CH
2
OH
Class 2
Ethyl ether
Diethyl ether
Ethoxyethane
1,1’-Oxybisethane
CH
3
CH
2
OCH
2
CH
3
Class 3
Ethyl formate
Formic acid ethyl ester
HCOOCH
2
CH
3
Class 3
Formamide
Methanamide
HCONH
2
Class 2
Formic acid
HCOOH
Class 3
Heptane
n-Heptane
CH
3
(CH
2
)
5
CH
3
Class 3
Hexane
n-Hexane
CH
3
(CH
2
)
4
CH
3
Class 2
Is obutyl acetate
Acetic acid isobutyl ester
CH
3
COOCH
2
CH(CH
3
)
2
Class 3
Isopropyl acetate
Acetic acid isopropyl ester
CH
3
COOCH(CH
3
)
2
Class 3
Methanol
Methyl alcohol
CH
3
OH
Class 2
2-Methoxyethanol
Methyl Cellos olve
CH
3
OCH
2
CH
2
OH
Class 2
Methyl acetate
Acetic acid methyl ester
CH
3
COOCH
3
Class 3
3-Methyl-1-butanol
Isoamyl alcohol
Isopentyl alcohol
3-Methylbutan-1-ol
(CH
3
)
2
CHCH
2
CH
2
OH
Class 3
3

Contains Nonbinding Recommendations
Methylbutyl ketone
2-Hexanone
Hexan-2-one
CH
3
(CH
2
)
3
COCH
3
Class 2
Methylcyclohexane
Cyclohexylmethane
Class 2
Methylethyl ketone
2-Butanone
MEK
Butan-2-one
CH
3
CH
2
COCH
3
Class 3
Methylisobutyl ketone
4-Methylpentan-2-one
4-Methyl-2-pentanone
MIBK
CH
3
COCH
2
CH(CH
3
)
2
Class 2
2-Methyl-1-propanol
Isobutyl alcohol
2-Methylpropan-1-ol
(CH
3
)
2
CHCH
2
OH
Class 3
N-Methylpyrrolidone
1-Methylpyrrolidin-2-one
1-Methyl-2-pyrrolidinone
Class 2
Nitromethane
CH
3
NO
2
Class 2
Pentane
n-Pentane
CH
3
(CH
2
)
3
CH
3
Class 3
1-Pentanol
Amyl alcohol
Pentan-1-ol
Pentyl alcohol
CH
3
(CH
2
)
3
CH
2
OH
Class 3
1-Propanol
Propan-1-ol
Propyl alcohol
CH
3
CH
2
CH
2
OH
Class 3
2-Propanol
Propan-2-ol
Isopropyl alcohol
(CH
3
)
2
CHOH
Class 3
Propyl acetate
Acetic acid propyl ester
CH
3
COOCH
2
CH
2
CH
3
Class 3
Pyridine
Class 2
Sulfolane
Tetrahydrothiophene 1,1-dioxide
Class 2
Tetrahydrofuran
Tetramethylene oxide
Oxacyclopentane
Class 2
Tetralin
1,2,3,4-Tetrahydro-naphthalene
Class 2
Toluene
Methylbenzene
Class 2
1,1,1-Trichloroethane
Methylchloroform
CH
3
CCl
3
Class 1
1,1,2-Trichloroethene
Trichloroethene
HClC=CCl
2
Class 2
Triethylamine
N,N-Diethylethanamine
N(CH
2
CH
3
)
3
Class 3
4
Contains Nonbinding Recommendations
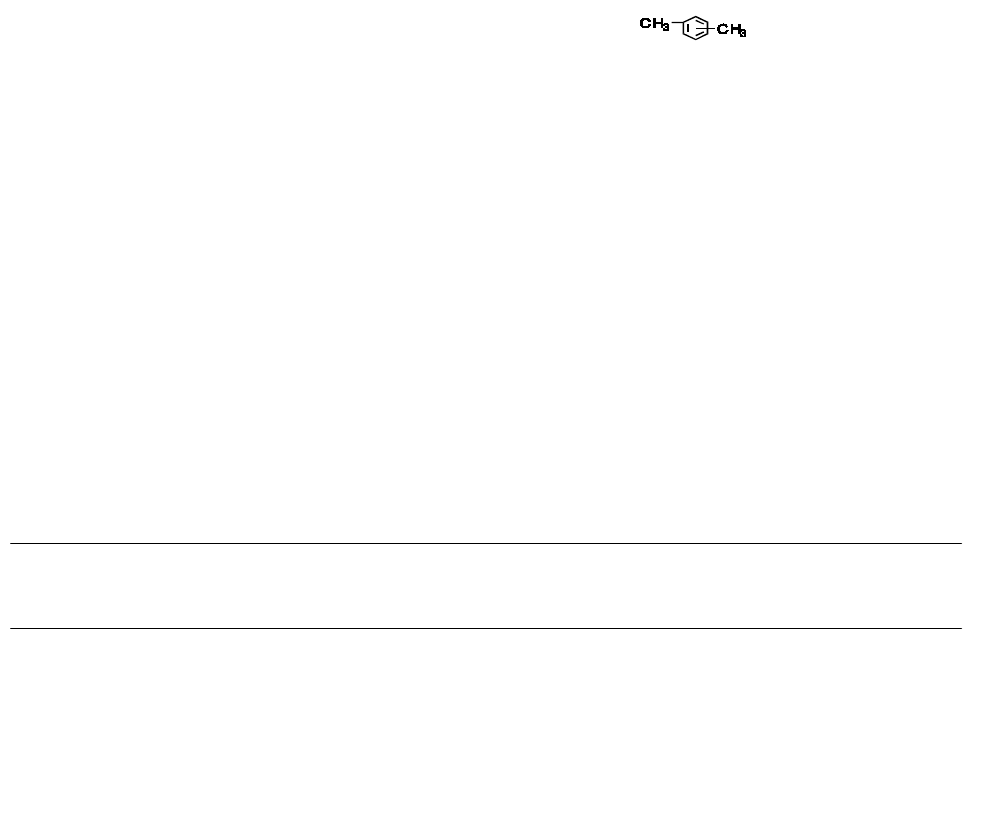
Xylene
1
Dimethybenzene
Xylol
Class 2
1
Us ually 60% m-xylene, 14% p-xylene, 9% o-xylene with 17% ethyl benzene.
III. SOLVENTS GROUPED BY CLASS
Solvents in Class 1 (Table 1) should not be employed in the manufacture of drug substances,
excipients, and drug products because of their unacceptable toxicity or their deleterious
environmental effect. However, if their use is unavoidable in order to produce a drug product
with a significant therapeutic advance, then their levels should be restricted as shown in Table 1,
unless otherwise justified. The solvent 1,1,1-Trichloroethane is included in Table 1 because it is
an environmental hazard. The stated limit of 1,500 ppm is based on a review of the safety data.
Table 1. – Class 1 Solvents in Pharmaceutical Products (Solvents That Should Be Avoided)
Solvent
Concentration Limit
(ppm)
Concern
Benzene
Carbon tetrachloride
2
4
Carcinogen
Toxic and environmental hazard
1,2-Dichloroethane
5
Toxic
1,1-Dichloroethene
8
Toxic
1,1,1-Trichloroethane 1,500 Environmental hazard
5

Contains Nonbinding Recommendations
Solvents in Class 2 (Table 2) should be limited in pharmaceutical products because of their
inherent toxicity. PDEs are given to the nearest 0.1 mg/day, and concentrations are given to the
nearest 10 ppm. The stated values do not reflect the necessary analytical precision of
determination. Precision should be determined as part of the validation of the method.
Table 2. – Class 2 Solvents in Pharmaceutical Products
Solvent
PDE (mg/day)
Concentration Limit (ppm)
Acetonitrile
4.1
410
Chlorobenzene
3.6
360
Chloroform
0.6
60
Cyclohexane
38.8
3,880
Cumene
0.7
70
1,2-Dichloroethene
18.7
1,870
Dichloromethane
6.0
600
1,2-Dimethoxyethane
1.0
100
N,N-Dimethylacetamide
10.9
1,090
N,N-Dimethylformamide
8.8
880
1,4-Dioxane
3.8
380
2-Ethoxyethanol
1.6
160
Ethyleneglycol
6.2
620
Formamide
2.2
220
Hexane
2.9
290
Methanol
30.0
3,000
2-Methoxyethanol
0.5
50
Methylbutyl ketone
0.5
50
Methylcyclohexane
11.8
1,180
Methylisobutylketone
2
45
4,500
N-Methylpyrrolidone
5.3
530
Nitromethane
0.5
50
Pyridine
2.0
200
Sulfolane
1.6
160
Tetrahydrofuran
7.2
720
Tetralin
1.0
100
2
The information included for Methylisobutylketone reflects that included in the Revision of PDE Information for
Methylisobutylketone, which reached Step 4 in November 2016 and was subsequently incorporated into the core
guidance.
6

Contains Nonbinding Recommendations
Toluene
8.9
890
1,1,2-Trichloroethene
0.8
80
Xylene
1
21.7
2,170
1
Us ually 60% m-xylene, 14% p-xylene, 9% o-xylene with 17% ethyl benzene.
Solvents in Class 3 (Table 3) may be regarded as less toxic and of lower risk to human health.
Class 3 includes no solvent known as a human health hazard at levels normally accepted in
pharmaceuticals. However, there are no long-term toxicity or carcinogenicity studies for many
of the solvents in Class 3. Available data indicate that they are less toxic in acute or short-term
studies and negative in genotoxicity studies. It is considered that amounts of these residual
solvents of 50 mg per day or less (corresponding to 5,000 ppm or 0.5 percent under Option 1)
would be acceptable without justification. Higher amounts may also be acceptable provided they
are realistic in relation to manufacturing capability and good manufacturing practice (GMP).
Table 3. – Class 3 Solvents Which Should Be Limited by GMP or Other Quality-Based
Requirements
Acetic acid
Heptane
Acetone Is obutyl acetate
Anisole Isopropyl acetate
1-Butanol Methyl acetate
2-Butanol 3-Methyl-1-butanol
Butyl acetate Methylethyl ketone
tert-Butylmethyl ether 2-Methyl-1-propanol
Dimethyl sulfoxide Pentane
Ethanol 1-Pentanol
Ethyl acetate 1-Propanol
Ethyl ether 2-Propanol
Ethyl formate Propyl acetate
Formic acid
Triethylamine
3
3
The information included for Triethylamine reflects that included in the Revision of PDE Information for
Triethylamine, which reached Step 4 in November 2016 and was subsequently incorporated into the core guidance.
7

Contains Nonbinding Recommendations
The solvents listed in Table 4 may also be of interest to manufacturers of excipients, drug
substances, or drug products. However, no adequate toxicological data on which to base a PDE
were found. Manufacturers should supply justification for residual levels of these solvents in
pharmaceutical products.
Table 4. – Solvents for Which No Adequate Toxicological Data Were Found
1,1-Diethoxypropane
Methylisopropyl ketone
1,1-Dimethoxymethane Methyltetrahydrofuran
2,2-Dimethoxypropane
Petroleum ether
Isooctane
Trichloroacetic acid
Isopropyl ether
Trifluoroacetic acid
8
