
ARAVA
®
Tablets
(leflunomide)
10 mg, 20 mg, 100 mg
Rx only
CONTRAINDICATIONS AND WARNINGS
Pregnancy
Pregnancy must be excluded before the start of treatment with ARAVA. ARAVA is
contraindicated in pregnant women, or women of childbearing potential who are not using
reliable contraception. (see CONTRAINDICATIONS and WARNINGS.) Pregnancy must
be avoided during ARAVA treatment or prior to the completion of the drug elimination
procedure after ARAVA treatment.
Hepatotoxicity
Severe liver injury, including fatal liver failure, has been reported in some patients treated
with ARAVA. Patients with pre-existing acute or chronic liver disease, or those with serum
alanine aminotransferase (ALT) >2xULN before initiating treatment, should not be treated
with ARAVA. Use caution when ARAVA is given with other potentially hepatotoxic drugs.
Monitoring of ALT levels is recommended at least monthly for six months after starting
ARAVA, and thereafter every 6-8 weeks. If ALT elevation > 3 fold ULN occurs, interrupt
ARAVA therapy while investigating the probable cause of the ALT elevation by close
observation and additional tests. If likely leflunomide-induced, start cholestyramine
washout and monitor liver tests weekly until normalized. If leflunomide-induced liver
injury is unlikely because some other probable cause has been found, resumption of
ARAVA therapy may be considered. (SEE WARNINGS – HEPATOTOXICITY).
DESCRIPTION
ARAVA
®
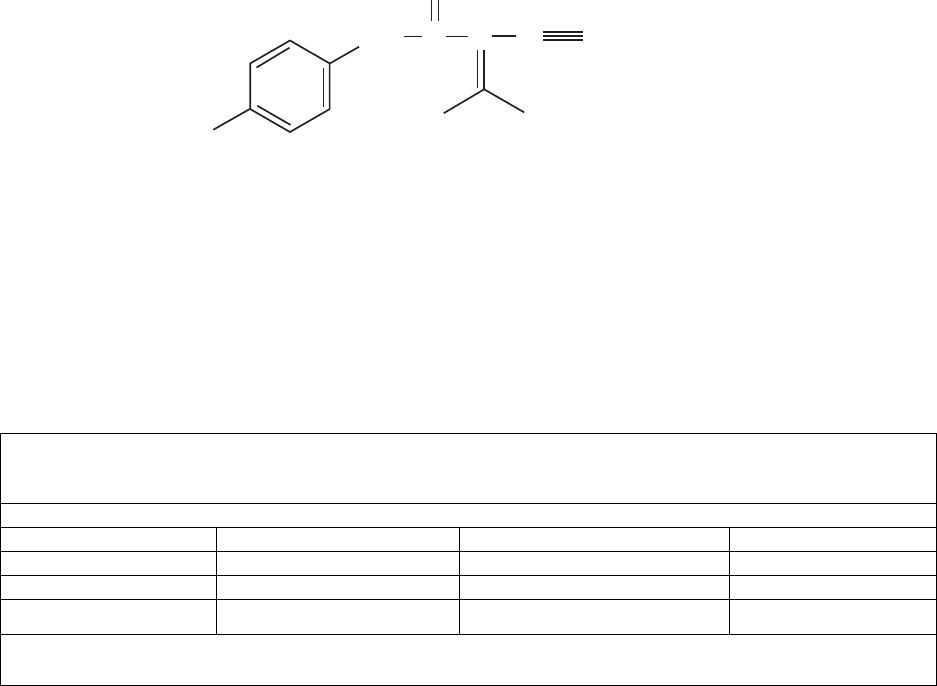
(leflunomide) is a pyrimidine synthesis inhibitor. The chemical name for leflunomide
is N-(4´-trifluoromethylphenyl)-5-methylisoxazole-4-carboxamide. It has an empirical formula
C
12
H
9
F
3
N
2
O
2
, a molecular weight of 270.2 and the following structural formula:
O
N
CH
3
CO
NH
CF
3
ARAVA is available for oral administration as tablets containing 10, 20, or 100 mg of active
drug. Combined with leflunomide are the following inactive ingredients: colloidal silicon
dioxide, crospovidone, hypromellose, lactose monohydrate, magnesium stearate, polyethylene
glycol, povidone, starch, talc, titanium dioxide, and yellow ferric oxide (20 mg tablet only).
1
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
CLINICAL PHARMACOLOGY
Mechanism of Action
Leflunomide is an isoxazole immunomodulatory agent which inhibits dihydroorotate
dehydrogenase (an enzyme involved in de novo pyrimidine synthesis) and has antiproliferative
activity. Several in vivo and in vitro experimental models have demonstrated an anti-
inflammatory effect.
Pharmacokinetics
Following oral administration, leflunomide is metabolized to an active metabolite A77 1726
(hereafter referred to as M1) which is responsible for essentially all of its activity in vivo.
Plasma levels of leflunomide are occasionally seen, at very low levels. Studies of the
pharmacokinetics of leflunomide have primarily examined the plasma concentrations of this
active metabolite.
O
NH
F
3
C
CH
3
C
C
C
N
OH
A77 1726 (M1)
Absorption
Following oral administration, peak levels of the active metabolite, M1, occurred between 6 - 12
hours after dosing. Due to the very long half-life of M1 (~2 weeks), a loading dose of 100 mg for
3 days was used in clinical studies to facilitate the rapid attainment of steady-state levels of M1.
Without a loading dose, it is estimated that attainment of steady-state plasma concentrations
would require nearly two months of dosing. The resulting plasma concentrations following both
loading doses and continued clinical dosing indicate that M1 plasma levels are dose proportional.
Table 1. Pharmacokinetic Parameters for M1 after Administration of Leflunomide at Doses of 5, 10,
and 25 mg/day for 24 Weeks to Patients (n=54) with Rheumatoid Arthritis (Mean ± SD)
(Study YU204)
Maintenance (Loading) Dose
Parameter 5 mg (50 mg) 10 mg (100 mg) 25 mg (100 mg)
C
24
(Day 1) (µg/mL)
1
4.0 ± 0.6 8.4 ± 2.1 8.5 ± 2.2
C
24
(ss) (µg/mL)
2
8.8 ± 2.9 18 ± 9.6 63 ± 36
t
1/2
(DAYS) 15 ± 3 14 ± 5 18 ± 9
1
Concentration at 24 hours after loading dose
2
Concentration at 24 hours after maintenance doses at steady state
Relative to an oral solution, ARAVA tablets are 80% bioavailable. Co-administration of
leflunomide tablets with a high fat meal did not have a significant impact on M1 plasma levels.
2
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
Distribution
M1 has a low volume of distribution (Vss = 0.13 L/kg) and is extensively bound (>99.3%) to
albumin in healthy subjects. Protein binding has been shown to be linear at therapeutic
concentrations. The free fraction of M1 is slightly higher in patients with rheumatoid arthritis
and approximately doubled in patients with chronic renal failure; the mechanism and
significance of these increases are unknown.
Metabolism
Leflunomide is metabolized to one primary (M1) and many minor metabolites. Of these minor
metabolites, only 4-trifluoromethylaniline (TFMA) is quantifiable, occurring at low levels in the
plasma of some patients. The parent compound is rarely detectable in plasma. At the present time
the specific site of leflunomide metabolism is unknown. In vivo and in vitro studies suggest a
role for both the GI wall and the liver in drug metabolism. No specific enzyme has been
identified as the primary route of metabolism for leflunomide; however, hepatic cytosolic and
microsomal cellular fractions have been identified as sites of drug metabolism.
Elimination
The active metabolite M1 is eliminated by further metabolism and subsequent renal excretion as
well as by direct biliary excretion. In a 28 day study of drug elimination (n=3) using a single
dose of radiolabeled compound, approximately 43% of the total radioactivity was eliminated in
the urine and 48% was eliminated in the feces. Subsequent analysis of the samples revealed the
primary urinary metabolites to be leflunomide glucuronides and an oxanilic acid derivative of
M1. The primary fecal metabolite was M1. Of these two routes of elimination, renal elimination
is more significant over the first 96 hours after which fecal elimination begins to predominate. In
a study involving the intravenous administration of M1, the clearance was estimated to be
31 mL/hr.
In small studies using activated charcoal (n=1) or cholestyramine (n=3) to facilitate drug
elimination, the in vivo plasma half-life of M1 was reduced from >1 week to approximately 1
day. (See PRECAUTIONS - General - Need for Drug Elimination). Similar reductions in
plasma half-life were observed for a series of volunteers (n=96) enrolled in pharmacokinetic
trials who were given cholestyramine. This suggests that biliary recycling is a major contributor
to the long elimination half-life of M1. Studies with both hemodialysis and CAPD (chronic
ambulatory peritoneal dialysis) indicate that M1 is not dialyzable.
Special Populations
Gender. Gender has not been shown to cause a consistent change in the in vivo
pharmacokinetics of M1.
Age. Age has been shown to cause a change in the in vivo pharmacokinetics of M1 (see
CLINICAL PHARMACOLOGY – Special Populations - Pediatrics).
Smoking. A population based pharmacokinetic analysis of the phase III data indicates that
smokers have a 38% increase in clearance over non-smokers; however, no difference in clinical
efficacy was seen between smokers and nonsmokers.
Chronic Renal Insufficiency. In single dose studies in patients (n=6) with chronic renal
insufficiency requiring either chronic ambulatory peritoneal dialysis (CAPD) or hemodialysis,
neither had a significant impact on circulating levels of M1. The free fraction of M1 was almost
3
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

doubled, but the mechanism of this increase is not known. In light of the fact that the kidney
plays a role in drug elimination and without adequate studies of leflunomide use in subjects with
renal insufficiency, caution should be used when ARAVA is administered to these patients.
Hepatic Insufficiency. Studies of the effect of hepatic insufficiency on M1 pharmacokinetics
have not been done. Given the need to metabolize leflunomide into the active species, the role of
the liver in drug elimination/recycling, and the possible risk of increased hepatic toxicity, the use
of leflunomide in patients with hepatic insufficiency is not recommended.
Pediatrics
The pharmacokinetics of M1 following oral administration of leflunomide have been
investigated in 73 pediatric patients with polyarticular course Juvenile Rheumatoid Arthritis
(JRA) who ranged in age from 3 to 17 years. The results of a population pharmacokinetic
analysis of these trials have demonstrated that pediatric patients with body weights ≤40 kg have
a reduced clearance of M1 (see Table 2) relative to adult rheumatoid arthritis patients.
Table 2: Population Pharmacokinetic Estimate of M1 Clearance Following Oral Administration of
Leflunomide in Pediatric Patients with Polyarticular Course JRA Mean ±SD [Range]
N Body Weight (kg) CL (mL/h)
10 <20
18 ± 9.8 [6.8-37]
30 20-40
18 ± 9.5 [4.2-43]
33 >40
26 ± 16 [9.7-93.6]
Drug Interactions
In vivo drug interaction studies have demonstrated a lack of a significant drug interaction
between leflunomide and tri-phasic oral contraceptives, and cimetidine.
In vitro studies of protein binding indicated that warfarin did not affect M1 protein binding. At
the same time M1 was shown to cause increases ranging from 13 - 50% in the free fraction of
diclofenac, ibuprofen and tolbutamide at concentrations in the clinical range. In vitro studies of
drug metabolism indicate that M1 inhibits CYP 450 2C9, which is responsible for the
metabolism of phenytoin, tolbutamide, warfarin and many NSAIDs. M1 has been shown to
inhibit the formation of 4´-hydroxydiclofenac from diclofenac in vitro. The clinical significance
of these findings with regard to phenytoin and tolbutamide is unknown; however, there was
extensive concomitant use of NSAIDs in the clinical studies and no differential effect was
observed. (See PRECAUTIONS – Drug Interactions).
Methotrexate. Coadministration, in 30 patients, of ARAVA (100 mg/day x 2 days followed by
10 - 20 mg/day) with methotrexate (10 - 25 mg/week, with folate) demonstrated no
pharmacokinetic interaction between the two drugs. However, co-administration increased risk
of hepatotoxicity (see PRECAUTIONS - Drug Interactions – Hepatotoxic Drugs).
Rifampin. Following concomitant administration of a single dose of ARAVA to subjects
receiving multiple doses of rifampin, M1 peak levels were increased (~40%) over those seen
when ARAVA was given alone. Because of the potential for ARAVA levels to continue to
increase with multiple dosing, caution should be used if patients are to receive both ARAVA and
rifampin.
4
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

CLINICAL STUDIES
A. ADULTS
The efficacy of ARAVA in the treatment of rheumatoid arthritis (RA) was demonstrated in three
controlled trials showing reduction in signs and symptoms, and inhibition of structural damage.
In two placebo controlled trials, efficacy was demonstrated for improvement in physical
function.
1. Reduction of signs and symptoms
Relief of signs and symptoms was assessed using the American College of Rheumatology
(ACR)20 Responder Index, a composite of clinical, laboratory, and functional measures in
rheumatoid arthritis. An “ACR20 Responder” is a patient who had ≥ 20% improvement in both
tender and swollen joint counts and in 3 of the following 5 criteria: physician global assessment,
patient global assessment, functional ability measure [Modified Health Assessment
Questionnaire (MHAQ)], visual analog pain scale, and erythrocyte sedimentation rate or
C-reactive protein. An “ACR20 Responder at Endpoint” is a patient who completed the study
and was an ACR20 Responder at the completion of the study.
2. Inhibition of structural damage
Inhibition of structural damage compared to control was assessed using the Sharp Score (Sharp,
JT. Scoring Radiographic Abnormalities in Rheumatoid Arthritis, Radiologic Clinics of North
America, 1996; vol. 34, pp. 233-241), a composite score of X-ray erosions and joint space
narrowing in hands/wrists and forefeet.
3. Improvement in physical function
Improvement in physical function was assessed using the Health Assessment Questionnaire
(HAQ) and the Medical Outcomes Survey Short Form (SF-36).
In all Arava monotherapy studies, an initial loading dose of 100 mg per day for three days only
was used followed by 20 mg per day thereafter.
US301 Clinical Trial in Adults
Study US301, a 2 year study, randomized 482 patients with active RA of at least 6 months
duration to leflunomide 20 mg/day (n=182), methotrexate 7.5 mg/week increasing to 15
mg/week (n=182), or placebo (n=118). All patients received folate 1 mg BID. Primary analysis
was at 52 weeks with blinded treatment to 104 weeks.
Overall, 235 of the 508 randomized treated patients (482 in primary data analysis and an
additional 26 patients), continued into a second 12 months of double-blind treatment (98
leflunomide, 101 methotrexate, 36 placebo). Leflunomide dose continued at 20 mg/day and the
methotrexate dose could be increased to a maximum of 20 mg/week. In total, 190 patients (83
leflunomide, 80 methotrexate, 27 placebo) completed 2 years of double-blind treatment.
The rate and reason for withdrawal is summarized in Table 3.
Table 3: Withdrawals in US301
n(%) patients
Leflunomide Placebo Methotrexate
190 128 190
5
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

Withdrawals in Year-1
Lack of efficacy 33 (17.4) 70 (54.7) 50 (26.3)
Safety 44 (23.2) 12 (9.4) 22 (11.6)
Other
1
15 (7.9) 10 (7.8) 17 (9.0)
Total 92 (48.4) 92 (71.9) 89 (46.8)
Patients entering Year 2 98 36 101
Withdrawals in Year-2
Lack of efficacy 4 (4.1) 1 (2.8) 4 (4.0)
Safety 8 (8.2) 0 (0.0) 10 (9.9)
Other
1
3 (3.1) 8 (22.2) 7 (6.9)
Total 15 (15.3) 9 (25.0) 21 (20.8)
1
Includes: lost to follow up, protocol violation, noncompliance, voluntary withdrawal, investigator discretion.
MN301/303/305 Clinical Trial in Adults
Study MN301 randomized 358 patients with active RA to leflunomide 20 mg/day (n=133),
sulfasalazine 2.0 g/day (n=133), or placebo (n=92). Treatment duration was 24 weeks. An
extension of the study was an optional 6-month blinded continuation of MN301 without the
placebo arm, resulting in a 12-month comparison of leflunomide and sulfasalazine (study
MN303).
Of the 168 patients who completed 12 months of treatment in MN301 and MN303, 146 patients
(87%) entered a 1-year extension study of double blind active treatment (MN305;
60 leflunomide, 60 sulfasalazine, 26 placebo/ sulfasalazine). Patients continued on the same
daily dosage of leflunomide or sulfasalazine that they had been taking at the completion of
MN301/303. A total of 121 patients (53 leflunomide, 47 sulfasalazine, 21 placebo/sulfasalazine)
completed the 2 years of double-blind treatment.
Patient withdrawal data in MN301/303/305 is summarized in Table 4.
Table 4: Withdrawals in study MN301/303/305
n(%) patients
Leflunomide Placebo Sulfasalazine
133 92 133
Withdrawals in MN301 (Mo 0-6)
Lack of efficacy
Safety
Other
1
Total
10 (7.5)
19 (14.3)
8 (6.0)
37 (27.8)
29 (31.5)
6 (6.5)
6 (6.5)
41 (44.6)
14 (10.5)
25 (18.8)
11 (8.3)
50 (37.6)
Patients entering MN303 80 76
Withdrawals in MN303 (Mo 7-12)
Lack of efficacy
Safety
Other
1
4 (5.0)
2 (2.5)
3 (3.8)
2 (2.6)
5 (6.6)
1 (1.3)
6
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

Total 9 (11.3) 8 (10.5)
Patients entering MN305 60 60
Withdrawals in MN305 (Mo 13-24)
Lack of efficacy 0 (0.0) 3 (5.0)
Safety 6 (10.0) 8 (13.3)
Other
1
1 (1.7) 2 (3.3)
Total 7 (11.7) 13 (21.7)
1
Includes: lost to follow up, protocol violation, noncompliance, voluntary withdrawal, investigator discretion.
MN302/304 Clinical Trial in Adults
Study MN302 randomized 999 patients with active RA to leflunomide 20 mg/day (n=501) or
methotrexate at 7.5 mg/week increasing to 15 mg/week (n=498). Folate supplementation was
used in 10% of patients. Treatment duration was 52 weeks.
Of the 736 patients who completed 52 weeks of treatment in study MN302, 612 (83%) entered
the double-blind, 1-year extension study MN304 (292 leflunomide, 320 methotrexate). Patients
continued on the same daily dosage of leflunomide or methotrexate that they had been taking at
the completion of MN302. There were 533 patients (256 leflunomide, 277 methotrexate) who
completed 2 years of double-blind treatment.
Patient withdrawal data in MN302/304 is summarized in Table 5.
Table 5: Withdrawals in MN302/304
n(%) patients
Leflunomide Methotrexate
501 498
Withdrawals in MN302 (Year-1)
Lack of efficacy 37 (7.4) 15 (3.0)
Safety 98 (19.6) 79 (15.9)
Other
1
17 (3.4) 17 (3.4)
Total 152 (30.3) 111 (22.3)
Patients entering MN304 292 320
Withdrawals in MN304 (Year-2)
Lack of efficacy 13 (4.5) 9 (2.8)
Safety 11 (3.8) 22 (6.9)
Other
1
12 (4.1) 12 (3.8)
Total 36 (12.3) 43 (13.4)
1
Includes: lost to follow up, protocol violation, noncompliance, voluntary withdrawal, investigator discretion.
7
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

Clinical Trial Data
1. Signs and symptoms Rheumatoid Arthritis
The ACR20 Responder at Endpoint rates are shown in Figure 1. ARAVA was statistically
significantly superior to placebo in reducing the signs and symptoms of RA by the primary
efficacy analysis, ACR20 Responder at Endpoint, in study US301 (at the primary 12 months
endpoint) and MN301 (at 6 month endpoint). ACR20 Responder at Endpoint rates with ARAVA
treatment were consistent across the 6 and 12 month studies (41 - 49%). No consistent
differences were demonstrated between leflunomide and methotrexate or between leflunomide
and sulfasalazine. ARAVA treatment effect was evident by 1 month, stabilized by 3 - 6 months,
and continued throughout the course of treatment as shown in Figure 2.
Figure 1
% ACR 20 Responder at Endpoint
Trial US301 Trial MN301 Trial MN302
(12 months) (6months) (12 months)
L=
Leflunomide
, M=
Methotrexate
, P=
Placebo
, S=
Sulfasalazine
-20
-10
0
10
20
30
40
50
60
70
1
Percentage
L L LM MP S P
40.7
35.0
18.6
48.5
44.7
28.6
43.4
56.6
Comparisons 95%Confidence Interval p Value
US301 Leflunomide vs. Placebo (12, 32) <0.0001
Methotrexate vs. Placebo (8, 30) <0.0001
Leflunomide vs. Methotrexate (-4, 16) NS
MN301 Leflunomide vs. Placebo (7, 33) 0.0026
Sulfasalazine vs. Placebo (4, 29) 0.0121
Leflunomide vs. Sulfasalazine (-8, 16) NS
MN302 Leflunomide vs. Methotrexate (-19, -7) <0.0001
Figure 2
8
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
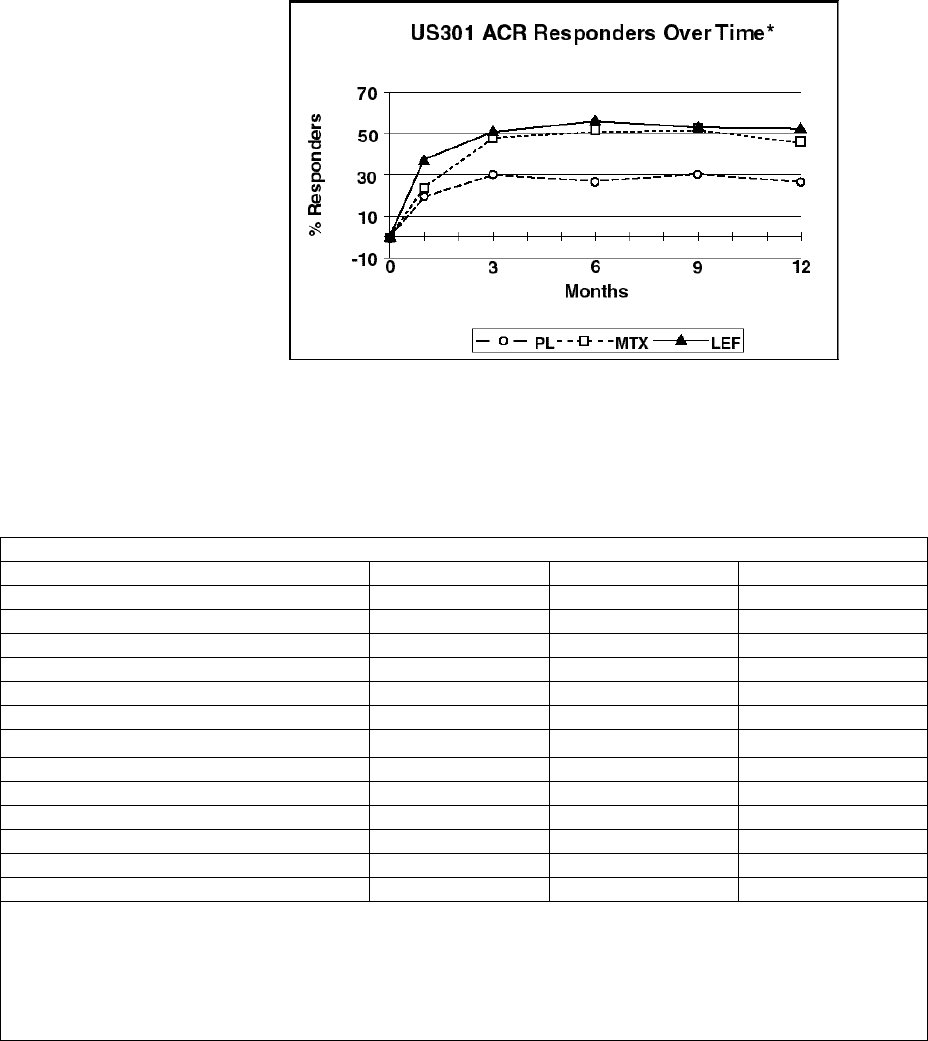
*Last Observation Carried Forward.
ACR50 and ACR70 Responders are defined in an analogous manner to the ACR 20 Responder,
but use improvements of 50% or 70%, respectively (Table 6). Mean change for the individual
components of the ACR Responder Index are shown in Table 7.
Table 6. Summary of ACR Response Rates*
Study and Treatment Group ACR20 ACR50 ACR70
Placebo-Controlled Studies
US301 (12 months)
Leflunomide (n=178)
†
52.2
‡
34.3
‡
20.2
‡
Placebo (n=118)
†
26.3 7.6 4.2
Methotrexate (n=180)
†
45.6 22.8 9.4
MN301(6 months)
Leflunomide (n=130)
†
54.6
‡
33.1
‡
10.0
§
Placebo (n=91)
†
28.6 14.3 2.2
Sulfasalazine (n=132)
†
56.8 30.3 7.6
Non-Placebo Active-Controlled Studies
MN302 (12 months)
Leflunomide (n=495)
†
51.1 31.1 9.9
Methotrexate (n=489)
†
65.2 43.8 16.4
* Intent to treat (ITT) analysis using last observation carried forward (LOCF) technique for patients who
discontinued early.
†
N is the number of ITT patients for whom adequate data were available to calculate the indicated rates.
‡
p<0.001 leflunomide vs placebo
§
p<0.02 leflunomide vs placebo
Table 7 shows the results of the components of the ACR response criteria for US301, MN301,
and MN302. ARAVA was significantly superior to placebo in all components of the ACR
Response criteria in study US301 and MN301. In addition, Arava was significantly superior to
placebo in improving morning stiffness, a measure of RA disease activity, not included in the
ACR Response criteria. No consistent differences were demonstrated between ARAVA and the
active comparators.
9
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

Table 7. Mean Change in the Components of the ACR Responder Index*
Components Placebo-Controlled Studies Non-placebo
Controlled Study
US301
(12 months)
MN301 Non-US
(6 months)
MN302 Non-US
(12 months)
Leflu-
nomide
Metho-
trexate
Placebo Leflu-
nomide
Sulfa-
salazine
Placebo Leflu-
nomide
Metho-
trexate
Tender joint count
1
-7.7 -6.6 -3.0 -9.7 -8.1 -4.3 -8.3 -9.7
Swollen joint count
1
-5.7 -5.4 -2.9 -7.2 -6.2 -3.4 -6.8 -9.0
Patient global
assessment
2
-2.1 -1.5 0.1 -2.8 -2.6 -0.9 -2.3 -3.0
Physician global
assessment
2
-2.8 -2.4 -1.0 -2.7 -2.5 -0.8 -2.3 -3.1
Physical
function/disability
(MHAQ/HAQ)
-0.29 -0.15 0.07 -0.50 -0.29 -0.04 -0.37 -0.44
Pain intensity
2
-2.2 -1.7 -0.5 -2.7 -2.0 -0.9 -2.1 -2.9
Erythrocyte
Sedimentation rate
-6.26 -6.48 2.56 -7.48 -16.56 3.44 -10.12 -22.18
C-reactive protein -0.62 -0.50 0.47 -2.26 -1.19 0.16 -1.86 -2.45
Not included in the ACR Responder Index
Morning Stiffness (min) -101.4 -88.7 14.7 -93.0 -42.4 -6.8 -63.7 -86.6
* Last Observation Carried Forward; Negative Change Indicates Improvement
1 Based on 28 joint count
2 Visual Analog Scale - 0=Best; 10=Worst
2. Maintenance of effect
After completing 12 months of treatment, patients continuing on study treatment were evaluated
for an additional 12 months of double-blind treatment (total treatment period of 2 years) in
studies US301, MN305, and MN304. ACR Responder rates at 12 months were maintained over
2 years in most patients continuing a second year of treatment.
Improvement from baseline in the individual components of the ACR responder criteria was also
sustained in most patients during the second year of Arava treatment in all three trials.
3. Inhibition of structural damage
The change from baseline to endpoint in progression of structural disease, as measured by the
Sharp X-ray score, is displayed in Figure 3. ARAVA was statistically significantly superior to
placebo in inhibiting the progression of disease by the Sharp Score. No consistent differences
were demonstrated between leflunomide and methotrexate or between leflunomide and
sulfasalazine.
Figure 3
10
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
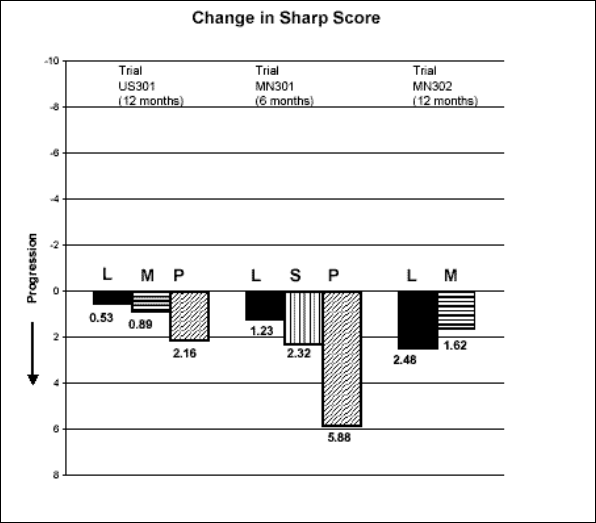
L= Leflunomide; M=methotrexate; S=sulfasalazine; P=placebo
Comparisons 95% Confidence Interval
p Value
US301 Leflunomide vs. Placebo (-4.0, -1.1) 0.0007
Methotrexate vs. Placebo (-2.6, -0.2) 0.0196
Leflunomide vs. Methotrexate (-2.3, 0.0) 0.0499
MN301 Leflunomide vs. Placebo (-6.2, -1.8) 0.0004
Sulfasalazine vs. Placebo (-6.9, 0.0) 0.0484
Leflunomide vs. Sulfasalazine (-3.3, 1.2) NS
MN302 Leflunomide vs. Methotrexate (-2.2, 7.4) NS
4. Improvement in physical function
The Health Assessment Questionnaire (HAQ) assesses a patient’s physical function and degree
of disability. The mean change from baseline in functional ability as measured by the HAQ
Disability Index (HAQ DI) in the 6 and 12 month placebo and active controlled trials is shown in
Figure 4. ARAVA was statistically significantly superior to placebo in improving physical
function. Superiority to placebo was demonstrated consistently across all eight HAQ DI
subscales (dressing, arising, eating, walking, hygiene, reach, grip and activities) in both placebo
controlled studies.
The Medical Outcomes Survey Short Form 36 (SF-36), a generic health-related quality of life
questionnaire, further addresses physical function. In US301, at 12 months, ARAVA provided
statistically significant improvements compared to placebo in the Physical Component Summary
(PCS) Score.
Figure 4
11
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
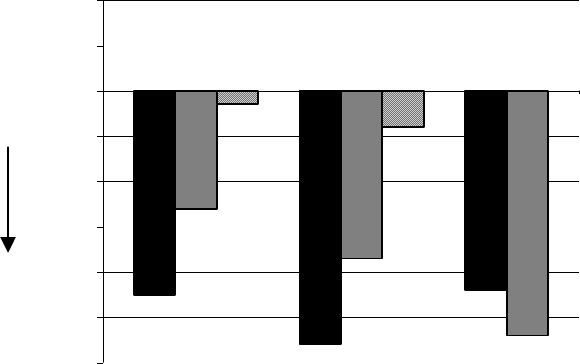
-0.6
-0.5
-0.4
-0.3
-0.2
-0.1
0
0.1
0.2
1
Improvement
L L LM MP PS
-0.45
-0.26
-0.03
-0.56
-0.37
-0.08
-0.44
-0.54
Trial
US301
(12 months)
Trial
MN301
(6
m
o
n
t
h
s)
Trial
MN302
(12 months)
Change in Functional Ability Measure*
* as measured by HAQ Disability Index
L
=Leflunomide,
M
=Methotrexate,
P
=Placebo,
S
=Sulfasalazine
Comparison 95% Confidence Interval p Value
US301 Leflunomide vs. Placebo (-0.58, -0.29) 0.0001
Leflunomide vs. Methotrexate (-0.34, -0.07) 0.0026
MN301 Leflunomide vs. Placebo (-0.67, -0.36) <0.0001
Leflunomide vs. Sulfasalazine (-0.33, -0.03) 0.0163
MN302 Leflunomide vs. Methotrexate (0.01, 0.16) 0.0221
Maintenance of effect
The improvement in physical function demonstrated at 6 and 12 months was maintained over
two years. In those patients continuing therapy for a second year, this improvement in physical
function as measured by HAQ and SF-36 (PCS) was maintained.
B. PEDIATRICS
Clinical Trials in Pediatrics
ARAVA was studied in a single multicenter, double-blind, active-controlled trial in 94 patients
(1:1 randomization) with polyarticular course juvenile rheumatoid arthritis (JRA) as defined by
the American College of Rheumatology (ACR). Approximately 68% of pediatric patients
receiving ARAVA, versus 89% of pediatric patients receiving the active comparator, improved
by Week 16 (end-of-study) employing the JRA Definition of Improvement (DOI) ≥ 30 %
responder endpoint. In this trial, the loading dose and maintenance dose of ARAVA was based
on three weight categories: <20 kg, 20-40kg, and >40 kg. The response rate to ARAVA in
pediatric patients ≤40 kg was less robust than in pediatric patients >40 kg suggesting suboptimal
dosing in smaller weight pediatric patients, as studied, resulting in less than efficacious plasma
concentrations, despite reduced clearance of M1. (See Pharmacokinetics - Pediatrics).
INDICATIONS AND USAGE
ARAVA is indicated in adults for the treatment of active rheumatoid arthritis (RA):
12
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
1. to reduce signs and symptoms
2. to inhibit structural damage as evidenced by X-ray erosions and joint space narrowing
3. to improve physical function.
(see CLINICAL STUDIES)
Aspirin, nonsteroidal anti-inflammatory agents and/or low dose corticosteroids may be continued
during treatment with ARAVA (see PRECAUTIONS – Drug Interactions – NSAIDs). The
combined use of ARAVA with antimalarials, intramuscular or oral gold, D penicillamine,
azathioprine, or methotrexate has not been adequately studied. (See WARNINGS -
Immunosuppression Potential/Bone Marrow Suppression).
CONTRAINDICATIONS
ARAVA is contraindicated in patients with known hypersensitivity to leflunomide or any of the
other components of ARAVA.
ARAVA can cause fetal harm when administered to a pregnant woman. Leflunomide, when
administered orally to rats during organogenesis at a dose of 15 mg/kg, was teratogenic (most
notably anophthalmia or microophthalmia and internal hydrocephalus). The systemic exposure
of rats at this dose was approximately 1/10 the human exposure level based on AUC. Under
these exposure conditions, leflunomide also caused a decrease in the maternal body weight and
an increase in embryolethality with a decrease in fetal body weight for surviving fetuses. In
rabbits, oral treatment with 10 mg/kg of leflunomide during organogenesis resulted in fused,
dysplastic sternebrae. The exposure level at this dose was essentially equivalent to the maximum
human exposure level based on AUC. At a 1 mg/kg dose, leflunomide was not teratogenic in rats
and rabbits.
When female rats were treated with 1.25 mg/kg of leflunomide beginning 14 days before mating
and continuing until the end of lactation, the offspring exhibited marked (greater than 90%)
decreases in postnatal survival. The systemic exposure level at 1.25 mg/kg was approximately
1/100 the human exposure level based on AUC.
ARAVA is contraindicated in women who are or may become pregnant. If this drug is used
during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be
apprised of the potential hazard to the fetus.
WARNINGS
Hepatotoxicity
Severe liver injury, including fatal liver failure, has been reported in some patients treated with
ARAVA. Patients with pre-existing acute or chronic liver disease, or those with serum alanine
aminotransferase (ALT) >2xULN before initiating treatment, should not be treated with
ARAVA. Use caution when ARAVA is given with other potentially hepatotoxic drugs.
Monitoring of ALT levels is recommended at least monthly for six months after starting
ARAVA, and thereafter every 6-8 weeks. If ALT elevation > 3 fold ULN occurs, interrupt
ARAVA therapy while investigating the probable cause of the ALT elevation by close
observation and additional tests. If likely leflunomide-induced, start cholestyramine washout and
13
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

monitor liver tests weekly until normalized (see PRECAUTIONS - General - Need for Drug
Elimination). If leflunomide-induced liver injury is unlikely because some other probable
cause has been found, resumption of ARAVA therapy may be considered.
In addition, if ARAVA and methotrexate are given concomitantly, ACR guidelines for
monitoring methotrexate liver toxicity must be followed with ALT, AST, and serum albumin
testing monthly.
In clinical trials, ARAVA treatment as monotherapy or in combination with methotrexate was
associated with elevations of liver enzymes, primarily ALT and AST, in a significant number of
patients; these effects were generally reversible. Most transaminase elevations were mild (≤ 2-
fold ULN) and usually resolved while continuing treatment. Marked elevations (>3-fold ULN)
occurred infrequently and reversed with dose reduction or discontinuation of treatment. Table 8
shows liver enzyme elevations seen with monthly monitoring in clinical trials US301 and
MN301. It was notable that the absence of folate use in MN302 was associated with a
considerably greater incidence of liver enzyme elevation on methotrexate.
Table 8. Liver Enzyme Elevations >3-fold Upper Limits of Normal (ULN)
US301 MN301 MN302*
LEF PL MTX LEF PL SSZ LEF MTX
ALT (SGPT)
>3-fold ULN
(n %)
Reversed to ≤ 2-fold ULN:
8
(4.4)
8
3
(2.5)
3
5
(2.7)
5
2
(1.5)
2
1
(1.1)
1
2
(1.5)
2
13
(2.6)
12
83
(16.7)
82
Timing of Elevation
0-3 Months
4-6 Months
7-9 Months
10-12 Months
6
1
1
-
1
1
1
-
1
3
1
-
2
-
-
-
1
-
-
-
2
-
-
-
7
1
-
5
27
34
16
6
*Only 10% of patients in MN302 received folate. All patients in US301 received folate.
In a 6 month study of 263 patients with persistent active rheumatoid arthritis despite
methotrexate therapy, and with normal LFTs, leflunomide was added to a group of 130 patients
starting at 10 mg per day and increased to 20 mg as needed. An increase in ALT greater than or
equal to three times the ULN was observed in 3.8% of patients compared to 0.8% in 133 patients
continued on methotrexate with placebo added.
Immunosuppression Potential/Bone Marrow Suppression
ARAVA is not recommended for patients with severe immunodeficiency, bone marrow
dysplasia, or severe, uncontrolled infections. In the event that a serious infection occurs, it may
be necessary to interrupt therapy with ARAVA and administer cholestyramine or charcoal (see
PRECAUTIONS – General – Need for Drug Elimination). Medications like leflunomide that
have immunosuppression potential may cause patients to be more susceptible to infections,
including opportunistic infections, especially Pneumocystis jiroveci pneumonia, tuberculosis
(including extra-pulmonary tuberculosis), and aspergillosis. Severe infections including sepsis,
which may be fatal, have been reported in patients receiving ARAVA, especially Pneumocystis
jiroveci pneumonia and aspergillosis. Most of the reports were confounded by concomitant
immunosuppressant therapy and/or comorbid illness which, in addition to rheumatoid disease,
may predispose patients to infection.
14
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
There have been rare reports of pancytopenia, agranulocytosis and thrombocytopenia in patients
receiving ARAVA alone. These events have been reported most frequently in patients who
received concomitant treatment with methotrexate or other immunosuppressive agents, or who
had recently discontinued these therapies; in some cases, patients had a prior history of a
significant hematologic abnormality.
Patients taking ARAVA should have platelet, white blood cell count and hemoglobin or
hematocrit monitored at baseline and monthly for six months following initiation of therapy and
every 6- to 8 weeks thereafter. If used with concomitant methotrexate and/or other potential
immunosuppressive agents, chronic monitoring should be monthly. If evidence of bone marrow
suppression occurs in a patient taking ARAVA, treatment with ARAVA should be stopped, and
cholestyramine or charcoal should be used to reduce the plasma concentration of leflunomide
active metabolite (see PRECAUTIONS – General – Need for Drug Elimination).
In any situation in which the decision is made to switch from ARAVA to another anti-rheumatic
agent with a known potential for hematologic suppression, it would be prudent to monitor for
hematologic toxicity, because there will be overlap of systemic exposure to both compounds.
ARAVA washout with cholestyramine or charcoal may decrease this risk, but also may induce
disease worsening if the patient had been responding to ARAVA treatment.
Skin Reactions
Rare cases of Stevens-Johnson syndrome and toxic epidermal necrolysis have been reported in
patients receiving ARAVA. If a patient taking ARAVA develops any of these conditions,
ARAVA therapy should be stopped, and a drug elimination procedure is recommended. (See
PRECAUTIONS - General - Need for Drug Elimination).
Malignancy
The risk of malignancy, particularly lymphoproliferative disorders, is increased with the use of
some immunosuppression medications. There is a potential for immunosuppression with
ARAVA. No apparent increase in the incidence of malignancies and lymphoproliferative
disorders was reported in the clinical trials of ARAVA, but larger and longer-term studies would
be needed to determine whether there is an increased risk of malignancy or lymphoproliferative
disorders with ARAVA.
Use in Women of Childbearing Potential
There are no adequate and well-controlled studies evaluating ARAVA in pregnant women.
However, based on animal studies, leflunomide may increase the risk of fetal death or
teratogenic effects when administered to a pregnant woman (see CONTRAINDICATIONS).
Women of childbearing potential must not be started on ARAVA until pregnancy is excluded
and it has been confirmed that they are using reliable contraception. Before starting treatment
with ARAVA, patients must be fully counseled on the potential for serious risk to the fetus.
The patient must be advised that if there is any delay in onset of menses or any other reason to
suspect pregnancy, they must notify the physician immediately for pregnancy testing and, if
positive, the physician and patient must discuss the risk to the pregnancy. It is possible that
rapidly lowering the blood level of the active metabolite by instituting the drug elimination
15
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
procedure described below at the first delay of menses may decrease the risk to the fetus from
ARAVA.
Upon discontinuing ARAVA, it is recommended that all women of childbearing potential
undergo the drug elimination procedure described below. Women receiving ARAVA treatment
who wish to become pregnant must discontinue ARAVA and undergo the drug elimination
procedure described below which includes verification of M1 metabolite plasma levels less than
0.02 mg/L (0.02 µg/mL). Human plasma levels of the active metabolite (M1) less than
0.02 mg/L (0.02 µg/mL) are expected to have minimal risk based on available animal data.
Peripheral Neuropathy
Cases of peripheral neuropathy have been reported in patients receiving ARAVA. Most patients
recovered after discontinuation of ARAVA, but some patients had persistent symptoms. Age
older than 60 years, concomitant neurotoxic medications, and diabetes may increase the risk for
peripheral neuropathy. If a patient taking ARAVA develops a peripheral neuropathy, consider
discontinuing ARAVA therapy and performing the drug elimination procedure (see
WARNINGS – Drug Elimination Procedure).
Drug Elimination Procedure
The following drug elimination procedure is recommended to achieve non-detectable plasma
levels (less than 0.02 mg/L or 0.02 µg/mL) after stopping treatment with ARAVA:
1) Administer cholestyramine 8 grams 3 times daily for 11 days. (The 11 days do not
need to be consecutive unless there is a need to lower the plasma level rapidly.)
2) Verify plasma levels less than 0.02 mg/L (0.02 µg/mL) by two separate tests at
least 14 days apart. If plasma levels are higher than 0.02 mg/L, additional
cholestyramine treatment should be considered.
Without the drug elimination procedure, it may take up to 2 years to reach plasma M1 metabolite
levels less than 0.02 mg/L due to individual variation in drug clearance.
PRECAUTIONS
General
Need for Drug Elimination
The active metabolite of leflunomide is eliminated slowly from the plasma. In instances of any
serious toxicity from ARAVA, including hypersensitivity, use of a drug elimination procedure as
described in this section is highly recommended to reduce the drug concentration more rapidly
after stopping ARAVA therapy. If hypersensitivity is the suspected clinical mechanism, more
prolonged cholestyramine or charcoal administration may be necessary to achieve rapid and
sufficient clearance. The duration may be modified based on the clinical status of the patient.
Cholestyramine given orally at a dose of 8 g three times a day for 24 hours to three healthy
volunteers decreased plasma levels of M1 by approximately 40% in 24 hours and by 49 to 65%
in 48 hours.
16
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
Administration of activated charcoal (powder made into a suspension) orally or via nasogastric
tube (50 g every 6 hours for 24 hours) has been shown to reduce plasma concentrations of the
active metabolite, M1, by 37% in 24 hours and by 48% in 48 hours.
These drug elimination procedures may be repeated if clinically necessary.
Respiratory
Interstitial lung disease has been reported during treatment with leflunomide and has been
associated with fatal outcomes (see ADVERSE REACTIONS). The risk of its occurrence is
increased in patients with a history of interstitial lung disease. Interstitial lung disease is a
potentially fatal disorder, which may occur acutely at any time during therapy and has a variable
clinical presentation. New onset or worsening pulmonary symptoms, such as cough and dyspnea,
with or without associated fever, may be a reason for discontinuation of the therapy and for
further investigation as appropriate. If discontinuation of the drug is necessary, initiation of
wash-out procedures should be considered. (See WARNINGS – Drug Elimination Procedure).
Tuberculosis Reactivation
Prior to initiating immunomodulatory therapies, including Arava, patients should be screened for
latent tuberculosis infection with a tuberculin skin test. Arava has not been studied in patients
with a positive tuberculosis screen, and the safety of Arava in individuals with latent tuberculosis
infection is unknown. Patients testing positive in tuberculosis screening should be treated by
standard medical practice prior to therapy with Arava.
Renal Insufficiency
Single dose studies in dialysis patients show a doubling of the free fraction of M1 in plasma.
There is no clinical experience in the use of ARAVA in patients with renal impairment. Caution
should be used when administering this drug in this population.
Vaccinations
No clinical data are available on the efficacy and safety of vaccinations during ARAVA
treatment. Vaccination with live vaccines is, however, not recommended. The long half-life of
ARAVA should be considered when contemplating administration of a live vaccine after
stopping ARAVA.
Blood Pressure Monitoring
Blood pressure should be checked before start of leflunomide treatment and periodically
thereafter.
Information for Patients
The potential for increased risk of birth defects should be discussed with female patients of
childbearing potential. It is recommended that physicians advise women that they may be at
increased risk of having a child with birth defects if they are pregnant when taking ARAVA,
become pregnant while taking ARAVA, or do not wait to become pregnant until they have
stopped taking ARAVA and followed the drug elimination procedure (as described in
WARNINGS – Use In Women of Childbearing Potential – Drug Elimination
Procedure).
17
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
Patients should be advised of the possibility of rare, serious skin reactions. Patients should be
instructed to inform their physicians promptly if they develop a skin rash or mucous
membrane lesions.
Patients should be advised of the potential hepatotoxic effects of ARAVA and of the need for
monitoring liver enzymes. Patients should be instructed to notify their physicians if they
develop symptoms such as unusual tiredness, abdominal pain or jaundice.
Patients should be advised that they may develop a lowering of their blood counts and should
have frequent hematologic monitoring. This is particularly important for patients who are
receiving other immunosuppressive therapy concurrently with ARAVA, who have recently
discontinued such therapy before starting treatment with ARAVA, or who have had a history
of a significant hematologic abnormality. Patients should be instructed to notify their
physicians promptly if they notice symptoms of pancytopenia (such as easy bruising or
bleeding, recurrent infections, fever, paleness or unusual tiredness).
Patients should be informed about the early warning signs of interstitial lung disease and
asked to contact their physician as soon as possible if these symptoms appear or worsen
during therapy.
Laboratory Tests
Hematologic Monitoring
At minimum, patients taking ARAVA should have platelet, white blood cell count and
hemoglobin or hematocrit monitored at baseline and monthly for six months following initiation
of therapy and every 6 to 8 weeks thereafter.
Bone Marrow Suppression Monitoring
If used concomitantly with immunosuppressants such as methotrexate, chronic monitoring
should be monthly. (See WARNINGS - Immunosuppression Potential/Bone Marrow
Suppression).
Liver Enzyme Monitoring
At minimum, ALT (SGPT) must be performed at baseline and at least monthly for six months
after starting ARAVA, and thereafter every 6-8 weeks. In addition, if ARAVA and methotrexate
are given concomitantly, ACR guidelines for monitoring methotrexate liver toxicity must be
followed with ALT, AST, and serum albumin testing every month. (See WARNINGS –
Hepatotoxicity.)
Due to a specific effect on the brush border of the renal proximal tubule, ARAVA has a
uricosuric effect. A separate effect of hypophosphaturia is seen in some patients. These effects
have not been seen together, nor have there been alterations in renal function.
Drug Interactions
Cholestyramine and Charcoal
Administration of cholestyramine or activated charcoal in patients (n=13) and volunteers (n=96)
resulted in a rapid and significant decrease in plasma M1 (the active metabolite of leflunomide)
concentration (see PRECAUTIONS – General – Need for Drug Elimination).
18
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
Hepatotoxic Drugs
Increased side effects may occur when leflunomide is given concomitantly with hepatotoxic
substances. This is also to be considered when leflunomide treatment is followed by such drugs
without a drug elimination procedure. In a small (n=30) combination study of ARAVA with
methotrexate, a 2- to 3-fold elevation in liver enzymes was seen in 5 of 30 patients. All
elevations resolved, 2 with continuation of both drugs and 3 after discontinuation of leflunomide.
A >3-fold increase was seen in another 5 patients. All of these also resolved, 2 with continuation
of both drugs and 3 after discontinuation of leflunomide. Three patients met “ACR criteria” for
liver biopsy (1: Roegnik Grade I, 2: Roegnik Grade IIIa). No pharmacokinetic interaction was
identified (see CLINICAL PHARMACOLOGY).
NSAIDs
In in vitro studies, M1 was shown to cause increases ranging from 13 - 50% in the free fraction
of diclofenac and ibuprofen at concentrations in the clinical range. The clinical significance of
this finding is unknown; however, there was extensive concomitant use of NSAIDs in clinical
studies and no differential effect was observed.
Tolbutamide
In in vitro studies, M1 was shown to cause increases ranging from 13 - 50% in the free fraction
of tolbutamide at concentrations in the clinical range. The clinical significance of this finding is
unknown.
Rifampin
Following concomitant administration of a single dose of ARAVA to subjects receiving multiple
doses of rifampin, M1 peak levels were increased (~40%) over those seen when ARAVA was
given alone. Because of the potential for ARAVA levels to continue to increase with multiple
dosing, caution should be used if patients are to be receiving both ARAVA and rifampin.
Warfarin
Increased INR (International Normalized Ratio) when ARAVA and warfarin were co-
administered has been rarely reported.
Carcinogenesis, Mutagenesis, and Impairment of Fertility
No evidence of carcinogenicity was observed in a 2-year bioassay in rats at oral doses of
leflunomide up to the maximally tolerated dose of 6 mg/kg (approximately 1/40 the maximum
human M1 systemic exposure based on AUC). However, male mice in a 2-year bioassay
exhibited an increased incidence in lymphoma at an oral dose of 15 mg/kg, the highest dose
studied (1.7 times the human M1 exposure based on AUC). Female mice, in the same study,
exhibited a dose-related increased incidence of bronchoalveolar adenomas and carcinomas
combined beginning at 1.5 mg/kg (approximately 1/10 the human M1 exposure based on AUC).
The significance of the findings in mice relative to the clinical use of ARAVA is not known.
Leflunomide was not mutagenic in the Ames Assay, the Unscheduled DNA Synthesis Assay, or
in the HGPRT Gene Mutation Assay. In addition, leflunomide was not clastogenic in the in vivo
Mouse Micronucleus Assay nor in the in vivo Cytogenetic Test in Chinese Hamster Bone
Marrow Cells. However, 4-trifluoromethylaniline (TFMA), a minor metabolite of leflunomide,
was mutagenic in the Ames Assay and in the HGPRT Gene Mutation Assay, and was clastogenic
19
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
in the in vitro Assay for Chromosome Aberrations in the Chinese Hamster Cells. TFMA was not
clastogenic in the in vivo Mouse Micronucleus Assay nor in the in vivo Cytogenetic Test in
Chinese Hamster Bone Marrow Cells. Leflunomide had no effect on fertility in either male or
female rats at oral doses up to 4.0 mg/kg (approximately 1/30 the human M1 exposure based on
AUC).
Pregnancy
Pregnancy Category X (see CONTRAINDICATIONS section). Pregnancy Registry: To
monitor fetal outcomes of pregnant women exposed to leflunomide, health care providers are
encouraged to register such patients by calling 1-877-311-8972.
Nursing Mothers
ARAVA should not be used by nursing mothers. It is not known whether ARAVA is excreted in
human milk. Many drugs are excreted in human milk, and there is a potential for serious adverse
reactions in nursing infants from ARAVA. Therefore, a decision should be made whether to
proceed with nursing or to initiate treatment with ARAVA, taking into account the importance of
the drug to the mother.
Use in Males
Available information does not suggest that ARAVA would be associated with an increased risk
of male-mediated fetal toxicity. However, animal studies to evaluate this specific risk have not
been conducted. To minimize any possible risk, men wishing to father a child should consider
discontinuing use of ARAVA and taking cholestyramine 8 grams 3 times daily for 11 days.
Pediatric Use
The safety and effectiveness of ARAVA in pediatric patients with polyarticular course juvenile
rheumatoid arthritis (JRA) have not been fully evaluated. (See CLINICAL STUDIES and
ADVERSE REACTIONS).
Geriatric Use
Of the total number of subjects in controlled clinical (Phase III) studies of ARAVA, 234 subjects
were 65 years and over. No overall differences in safety or effectiveness were observed between
these subjects and younger subjects, and other reported clinical experience has not identified
differences in responses between the elderly and younger patients, but greater sensitivity of some
older individuals cannot be ruled out. No dosage adjustment is needed in patients over 65.
ADVERSE REACTIONS
Adverse reactions associated with the use of leflunomide in RA include diarrhea, elevated liver
enzymes (ALT and AST), alopecia and rash. In the controlled studies at one year, the following
adverse events were reported, regardless of causality. (See Table 9.)
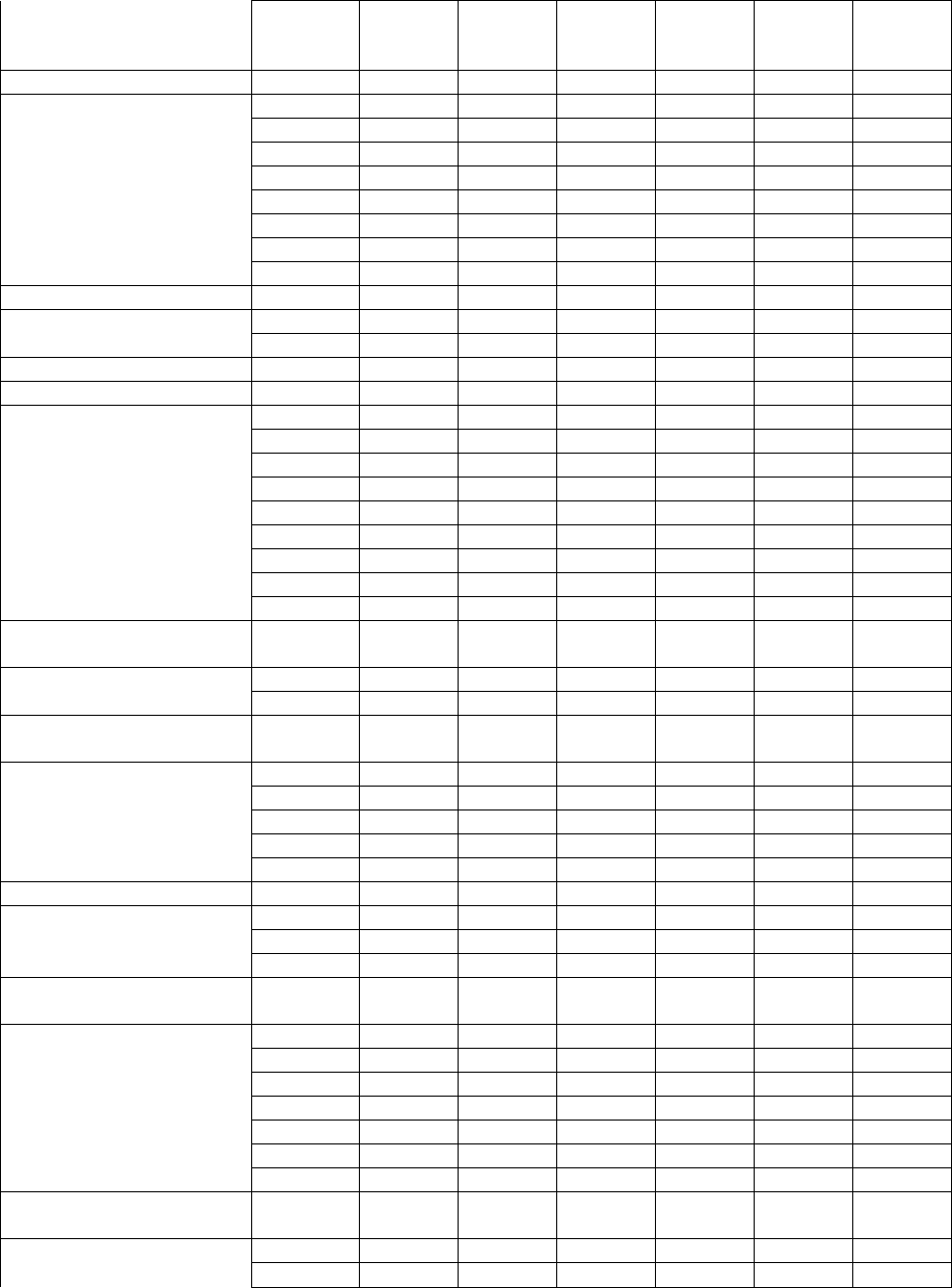
Table 9. Percentage Of Patients With Adverse Events ≥3% In Any Leflunomide Treated Group
All RA
Studies
Placebo-Controlled Trials Active-Controlled
Trials
MN 301 and US 301 MN 302*
20
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
LEF
(N=1339)
1
LEF
(N=315)
PBO
(N=210)
SSZ
(N=133)
MTX
(N=182)
LEF
(N=501)
MTX
(N=498)
BODY AS A WHOLE
Allergic Reaction 2% 5% 2% 0% 6% 1% 2%
Asthenia 3% 6% 4% 5% 6% 3% 3%
Flu Syndrome 2% 4% 2% 0% 7% 0% 0%
Infection, upper respiratory 4% 0% 0% 0% 0% 0% 0%
Injury Accident 5% 7% 5% 3% 11% 6% 7%
Pain 2% 4% 2% 2% 5% 1% <1%
Abdominal Pain 6% 5% 4% 4% 8% 6% 4%
Back Pain 5% 6% 3% 4% 9% 8% 7%
CARDIOVASCULAR
Hypertension
2
10% 9% 4% 4% 3% 10% 4%
- New onset of hypertension 1% <1% 0% 2% 2% <1%
Chest Pain 2% 4% 2% 2% 4% 1% 2%
GASTROINTESTINAL
Anorexia 3% 3% 2% 5% 2% 3% 3%
Diarrhea 17% 27% 12% 10% 20% 22% 10%
Dyspepsia 5% 10% 10% 9% 13% 6% 7%
Gastroenteritis 3% 1% 1% 0% 6% 3% 3%
Abnormal Liver Enzymes 5% 10% 2% 4%
10% 6% 17%
Nausea 9% 13% 11% 19% 18% 13% 18%
GI/Abdominal Pain 5% 6% 4% 7% 8% 8% 8%
Mouth Ulcer 3% 5% 4% 3% 10% 3% 6%
Vomiting 3% 5% 4% 4% 3% 3% 3%
METABOLIC AND
NUTRITIONAL
Hypokalemia
Weight Loss
3
1% 3% 1% 1% 1% 1% <1%
4% 2% 1% 2% 0% 2% 2%
MUSCULO-SKELETAL
SYSTEM
Arthralgia 1% 4% 3% 0% 9% <1% 1%
Leg Cramps 1% 4% 2% 2% 6% 0% 0%
Joint Disorder 4% 2% 2% 2% 2% 8% 6%
Synovitis 2% <1% 1% 0% 2% 4% 2%
Tenosynovitis 3% 2% 0% 1% 2% 5% 1%
NERVOUS SYSTEM
Dizziness 4% 5% 3% 6% 5% 7% 6%
Headache 7% 13% 11% 12% 21% 10% 8%
Paresthesia 2% 3% 1% 1% 2% 4% 3%
RESPIRATORY
SYSTEM
Bronchitis 7% 5% 2% 4% 7% 8% 7%
Increased Cough 3% 4% 5% 3% 6% 5% 7%
Respiratory Infection 15% 21% 21% 20% 32% 27% 25%
Pharyngitis 3% 2% 1% 2% 1% 3% 3%
Pneumonia 2% 3% 0% 0% 1% 2% 2%
Rhinitis 2% 5% 2% 4% 3% 2% 2%
Sinusitis 2% 5% 5% 0% 10% 1% 1%
SKIN AND
APPENDAGES
Alopecia 10% 9% 1% 6% 6% 17% 10%
Eczema 2% 1% 1% 1% 1% 3% 2%
21
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
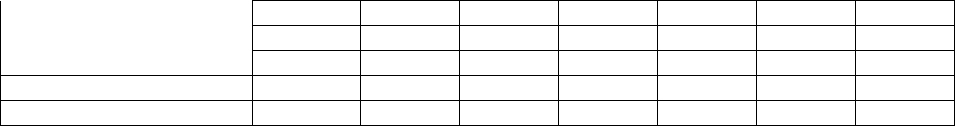
Pruritus 4% 5% 2% 3% 2% 6% 2%
Rash 10% 12% 7% 11% 9% 11% 10%
Dry Skin 2% 3% 2% 2% 0% 3% 1%
UROGENITAL SYSTEM
Urinary Tract Infection 5% 5% 7% 4% 2% 5% 6%
* Only 10% of patients in MN302 received folate. All patients in US301 received folate; none
in MN301 received folate.
1 Includes all controlled and uncontrolled trials with leflunomide (duration up to 12 months).
2 Hypertension as a preexisting condition was overrepresented in all leflunomide treatment
groups in phase III trials
3 In a meta-analysis of all phase II and III studies, during the first 6 months in patients
receiving leflunomide, 10% lost 10-19 lbs (24 cases per 100 patient years) and 2% lost at
least 20 lbs (4 cases/100 patient years). Of patients receiving leflunomide, 4% lost 10% of
their baseline weight during the first 6 months of treatment.
Adverse events during a second year of treatment with leflunomide in clinical trials were
consistent with those observed during the first year of treatment and occurred at a similar or
lower incidence.
In addition, the following adverse events have been reported in 1% to <3% of the RA patients in
the leflunomide treatment group in controlled clinical trials.
Body as a Whole: abscess, cyst, fever, hernia, malaise, pain, neck pain, pelvic pain;
Cardiovascular: angina pectoris, migraine, palpitation, tachycardia, varicose vein, vasculitis,
vasodilatation;
Gastrointestinal: cholelithiasis, colitis, constipation, esophagitis, flatulence, gastritis, gingivitis,
melena, oral moniliasis, pharyngitis, salivary gland enlarged, stomatitis (or aphthous stomatitis),
tooth disorder;
Endocrine: diabetes mellitus, hyperthyroidism;
Hemic and Lymphatic System: anemia (including iron deficiency anemia), ecchymosis;
Metabolic and Nutritional: creatine phosphokinase increased, hyperglycemia, hyperlipidemia,
peripheral edema;
Musculo-Skeletal System: arthrosis, bone necrosis, bone pain, bursitis, muscle cramps, myalgia,
tendon rupture;
Nervous System: anxiety, depression, dry mouth, insomnia, neuralgia, neuritis, sleep disorder,
sweating increased, vertigo;
Respiratory System: asthma, dyspnea, epistaxis, lung disorder;
Skin and Appendages: acne, contact dermatitis, fungal dermatitis, hair discoloration,
hematoma, herpes simplex, herpes zoster, maculopapular rash, nail disorder, skin discoloration,
skin disorder, skin nodule, subcutaneous nodule, ulcer skin;
Special Senses: blurred vision, cataract, conjunctivitis, eye disorder, taste perversion;
22
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
Urogenital System: albuminuria, cystitis, dysuria, hematuria, menstrual disorder, prostate
disorder, urinary frequency, vaginal moniliasis.
Other less common adverse events seen in clinical trials include: 1 case of anaphylactic reaction
occurred in Phase 2 following rechallenge of drug after withdrawal due to rash (rare); urticaria;
eosinophilia; transient thrombocytopenia (rare); and leukopenia <2000 WBC/mm
3
(rare).
Adverse events during a second year of treatment with leflunomide in clinical trials were
consistent with those observed during the first year of treatment and occurred at a similar or
lower incidence.
In post-marketing experience, the following have been reported rarely:
Body as a whole: opportunistic infections, severe infections including sepsis that may be fatal;
Gastrointestinal: pancreatitis;
Hematologic: agranulocytosis, leukopenia, neutropenia, pancytopenia, thrombocytopenia;
Hypersensitivity: angioedema;
Hepatic: hepatitis, jaundice/cholestasis, severe liver injury such as hepatic failure and acute
hepatic necrosis that may be fatal;
Respiratory: interstitial lung disease, including interstitial pneumonitis and pulmonary fibrosis,
which may be fatal;
Nervous system: peripheral neuropathy;
Skin and Appendages: erythema multiforme, Stevens-Johnson syndrome, toxic epidermal
necrolysis, vasculitis including cutaneous necrotizing vasculitis.
Adverse Reactions (Pediatric Patients)
The safety of ARAVA was studied in 74 patients with polyarticular course juvenile rheumatoid
arthritis ranging in age from 3-17 years (47 patients from the active-controlled study and 27 from
an open-label safety and pharmacokinetic study). The most common adverse events included
abdominal pain, diarrhea, nausea, vomiting, oral ulcers, upper respiratory tract infections,
alopecia, rash, headache, and dizziness. Less common adverse events included anemia,
hypertension, and weight loss. Fourteen pediatric patients experienced ALT and/or AST
elevations, nine between 1.2 and 3-fold the upper limit of normal, five between 3 and 8-fold the
upper limit of normal.
DRUG ABUSE AND DEPENDENCE
ARAVA has no known potential for abuse or dependence.
OVERDOSAGE
In mouse and rat acute toxicology studies, the minimally toxic dose for oral leflunomide was 200
- 500 mg/kg and 100 mg/kg, respectively (approximately >350 times the maximum
recommended human dose, respectively).
There have been reports of chronic overdose in patients taking ARAVA at daily dose up to five
times the recommended daily dose and reports of acute overdose in adults or children. There
23
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
were no adverse events reported in the majority of case reports of overdose. Adverse events were
consistent with the safety profile for ARAVA (see ADVERSE REACTIONS). The most
frequent adverse events observed were diarrhea, abdominal pain, leukopenia, anemia and
elevated liver function tests.
In the event of a significant overdose or toxicity, cholestyramine or charcoal administration is
recommended to accelerate elimination (see PRECAUTIONS – General – Need for Drug
Elimination).
Studies with both hemodialysis and CAPD (chronic ambulatory peritoneal dialysis) indicate that
M1, the primary metabolite of leflunomide, is not dialyzable. (See CLINICAL
PHARMACOLOGY – Elimination).
DOSAGE AND ADMINISTRATION
Loading Dose
Due to the long half-life in patients with RA and recommended dosing interval (24 hours), a
loading dose is needed to provide steady-state concentrations more rapidly. It is recommended
that ARAVA therapy be initiated with a loading dose of one 100 mg tablet per day for 3 days.
Elimination of the loading dose regimen may decrease the risk of adverse events. This could be
especially important for patients at increased risk of hematologic or hepatic toxicity, such as
those receiving concomitant treatment with methotrexate or other immunosuppressive agents or
on such medications in the recent past. (See WARNINGS - Hepatotoxicity).
Maintenance Therapy
Daily dosing of 20 mg is recommended for treatment of patients with RA. A small cohort of
patients (n=104), treated with 25 mg/day, experienced a greater incidence of side effects;
alopecia, weight loss, liver enzyme elevations. Doses higher than 20 mg/day are not
recommended. If dosing at 20 mg/day is not well tolerated clinically, the dose may be decreased
to 10 mg daily. Due to the prolonged half-life of the active metabolite of leflunomide, patients
should be carefully observed after dose reduction, since it may take several weeks for metabolite
levels to decline.
Monitoring
Hematology parameters and liver enzymes should be monitored (see PRECAUTIONS –
Laboratory Tests; WARNINGS – Hepatotoxicity; WARNINGS – Immunosuppression
Potential/Bone Marrow Suppression).
HOW SUPPLIED
ARAVA Tablets in 10 and 20 mg strengths are packaged in bottles. ARAVA Tablets 100 mg
strength are packaged in blister packs.
ARAVA
®
(leflunomide) Tablets
Strength Quantity NDC Number Description
10 mg 30 count bottle 0088-2160-30 White, round film-coated tablet embossed with
“ZBN” on one side.
20 mg 30 count bottle 0088-2161-30 Light yellow, triangular film-coated tablet
24
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda

embossed with “ZBO” on one side.
100 mg 3 count blister pack 0088-2162-33 White, round film-coated tablet embossed with
“ZBP” on one side.
Store at 25°C (77°F); excursions permitted to 15-30°C (59-86°F) [see USP Controlled Room
Temperature]. Protect from light.
Rx only.
Revised July 2011
sanofi-aventis U.S. LLC
Bridgewater, NJ 08807
©2011 sanofi-aventis U.S. LLC
25
Reference ID: 2971145
This label may not be the latest approved by FDA.
For current labeling information, please visit https://www.fda.gov/drugsatfda
