
_________________
______________
_____________
____________________ ___________________
_______________ _______________
_______________
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use
KRAZATI safely and effectively. See full prescribing information for
KRAZATI.
KRAZATI
™
(adagrasib) tablets, for oral use
Initial U.S. Approval: 2022
__________________
INDICATIONS AND USAGE
KRAZATI is an inhibitor of the RAS GTPase family indicated for the
treatment of adult patients with KRAS G12C-mutated locally advanced or
metastatic non-small cell lung cancer (NSCLC), as determined by an FDA
approved test, who have received at least one prior systemic therapy. (1)
This indication is approved under accelerated approval based on objective
response rate (ORR) and duration of response (DOR). Continued approval for
this indication may be contingent upon verification and description of a
clinical benefit in a confirmatory trial(s). (1)
_______________
DOSAGE AND ADMINISTRATION
• Recommended dosage: 600 mg orally twice daily. (2.2)
• Swallow tablets whole with or without food. (2.2)
______________
DOSAGE FORMS AND STRENGTHS
Tablets: 200 mg. (3)
CONTRAINDICATIONS
None. (4)
WARNINGS AND PRECAUTIONS
• Gastrointestinal Adverse Reactions: Monitor patients for diarrhea, nausea
and vomiting and provide supportive care as needed. Withhold, reduce the
dose or permanently discontinue based on severity. (2.3, 5.1)
• QTc Interval Prolongation: Avoid concomitant use of KRAZATI with
other products with a known potential to prolong the QTc interval.
Monitor ECG and electrolytes in patients at risk, and in patients taking
medications known to prolong the QT interval. Withhold, reduce the dose,
or permanently discontinue based on severity. (
2.3, 5.2)
• Hepatotoxicity: Monitor liver laboratory tests prior to the start of
KRAZATI and monthly for 3 months after and as clinically indicated.
Reduce the dose, withhold, or permanently discontinue based on severity.
(2.3, 5.3)
• Interstitial Lung Disease / Pneumonitis: Monitor for new or worsening
respiratory symptoms. Withhold KRAZATI for suspected
ILD/pneumonitis and permanently discontinue if no other potential causes
of ILD/pneumonitis are identified. (
2.3, 5.4)
____________________
ADVERSE REACTIONS
____________________
• The most common (≥ 25%) adverse reactions were nausea, diarrhea,
vomiting, fatigue, musculoskeletal pain, hepatotoxicity, renal impairment,
edema, dyspnea, and decreased appetite. (6.1)
• The most common Grade 3 or 4 (≥ 2%) laboratory abnormalities were
decreased lymphocytes, decreased hemoglobin, increased alanine
aminotransferase, increased aspartate aminotransferase, hypokalemia,
hyponatremia, increased lipase, decreased leukocytes, decreased
neutrophils and increased alkaline phosphatase. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Mirati
Therapeutics, Inc. at 1-844-MIRATI-1 (1-844-647-2841) or FDA at
1-800-FDA-1088 or www.fda.gov/medwatch.
____________________
DRUG INTERACTIONS
____________________
See full prescribing information for clinically significant drug interactions
with KRAZATI. (7)
• Strong CYP3A4 Inducers: Avoid concomitant use. (7.1
)
• Strong CYP3A4 Inhibitors: Avoid concomitant use until adagrasib
concentrations have reached steady state. (7.1)
• Sensitive CYP3A4 Substrates: Avoid concomitant use with sensitive
CYP3A4 substrates. (7.2)
• Sensitive CYP2C9 or CYP2D6 Substrates or P-gp Substrates: Avoid
concomitant use with sensitive CYP2C9 or CYP2D6 substrates or P-gp
substrates where minimal concentration changes may lead to serious
adverse reactions. (7.2)
• Drugs That Prolong QT Interval: Avoid concomitant use with KRAZATI.
(7.3)
USE IN SPECIFIC POPULATIONS
_______________
Lactation: Advise not to breastfeed. (8.2)
See 17 for PATIENT COUNSELING INFORMATION and FDA-
approved patient labeling.
Revised: 12/2022
FULL PRESCRIBING INFORMATION: CONTENTS*
1
INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Patient Selection
2.2 Recommended Dosage
2.3 Dosage Modifications for Adverse Reactions
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Gastrointestinal Adverse Reactions
5.2 QTc Interval Prolongation
5.3 Hepatotoxicity
5.4 Interstitial Lung Disease /Pneumonitis
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
7.1 Effects of Other Drugs on KRAZATI
7.2 Effects of KRAZATI on Other Drugs
7.3 Drugs That Prolong QTc Interval
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
13.2 Animal Toxicology and/or Pharmacology
14 CLINICAL STUDIES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
*Sections or subsections omitted from the full prescribing information are not
listed.
Reference ID: 5095255

FULL PRESCRIBING INFORMATION
1 INDICATIONS AND USAGE
KRAZATI is indicated for the treatment of adult patients with KRAS G12C-mutated locally
advanced or metastatic non-small cell lung cancer (NSCLC), as determined by an FDA-approved
test [see Dosage and Administration (2.1)], who have received at least one prior systemic
therapy.
This indication is approved under accelerated approval based on objective response rate (ORR)
and duration of response (DOR) [see Clinical Studies (14)]. Continued approval for this
indication may be contingent upon verification and description of a clinical benefit in a
confirmatory trial(s).
2 DOSAGE AND ADMINISTRATION
2.1 Patient Selection
Select patients for treatment of locally advanced or metastatic NSCLC with KRAZATI based on
the presence of KRAS G12C mutation in plasma or tumor specimens [see Clinical Studies (14)].
If no mutation is detected in a plasma specimen, test tumor tissue.
Information on FDA-approved tests for the detection of a KRAS G12C mutation is available at:
https://www.fda.gov/CompanionDiagnostics
2.2 Recommended Dosage
The recommended dosage of KRAZATI is 600 mg orally twice daily until disease progression or
unacceptable toxicity.
Take KRAZATI at the same time every day with or without food [see Clinical Pharmacology
(12.3)]. Swallow tablets whole. Do not chew, crush or split tablets.
If vomiting occurs after taking KRAZATI, do not take an additional dose. Resume dosing at the
next scheduled time.
If a dose is inadvertently missed, it should be skipped if greater than 4 hours have elapsed from
the expected dosing time. Resume dosing at the next scheduled time.
2.3 Dosage Modifications for Adverse Reactions
Recommended dose reductions for adverse reactions are outlined in Table 1. If adverse reactions
occur, a maximum of two dose reductions are permitted. Permanently discontinue KRAZATI in
patients who are unable to tolerate 600 mg once daily.
Reference ID: 5095255
2
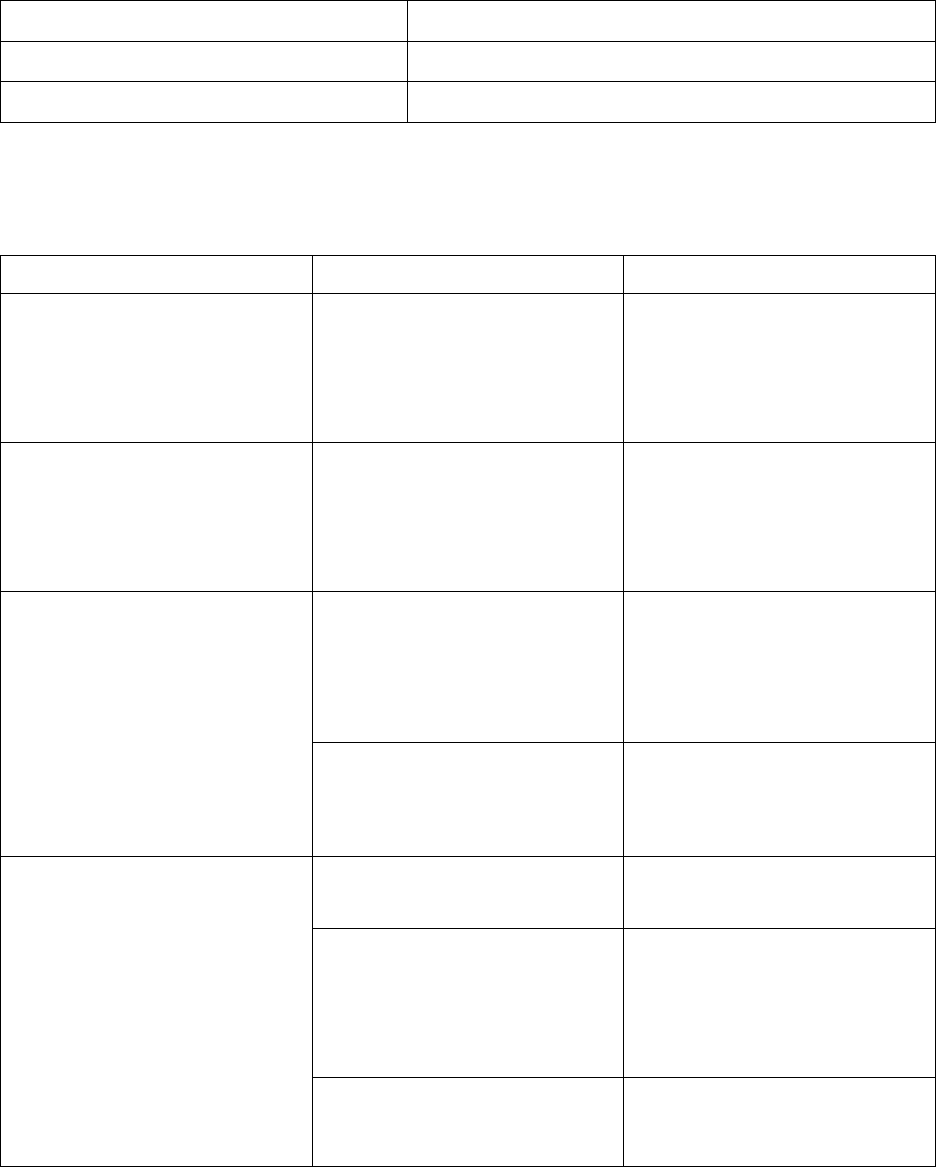
Table 1: Recommended KRAZATI Dosage Reductions for Adverse Reactions
Dose Reduction Dosage
First dose reduction 400 mg twice daily
Second dose reduction 600 mg once daily
The recommended dosage modifications for adverse reactions are provided in Table 2.
Table 2: Recommended KRAZATI Dosage Modifications for Adverse Reactions
Adverse reaction Severity
a
Dosage Modification
Nausea or vomiting despite
appropriate supportive care
(including anti-emetic therapy)
[see Warnings and Precautions
(5.1)]
Grade 3 or 4
• Withhold KRAZATI until
recovery to ≤ Grade 1 or
return to baseline.
• Resume KRAZATI at the
next lower dose level.
Diarrhea despite appropriate
supportive care (including anti-
diarrheal therapy)
[see Warnings and Precautions
(5.1)]
Grade 3 or 4
• Withhold KRAZATI until
recovery to ≤ Grade 1 or
return to baseline.
• Resume KRAZATI at the
next lower dose level.
QTc Interval Prolongation
[see Warnings and Precautions
(5.2)]
QTc absolute value greater than
500 ms
or
Greater than an increase of
60 ms from baseline
• Withhold KRAZATI until
QTc interval less than
481 ms or return to baseline.
• Resume KRAZATI at the
next lower dose level.
Torsade de pointes, polymorphic
ventricular tachycardia or signs
or symptoms of serious or life-
threatening arrhythmia
• Permanently discontinue
KRAZATI.
Hepatotoxicity
[see Warnings and Precautions
(5.3)]
Grade 2
AST or ALT
• Decrease KRAZATI to the
next lower dose level.
Grade 3 or 4
• Withhold KRAZATI until
AST or ALT
recovery to ≤ Grade 1 or
return to baseline.
• Resume KRAZATI at the
next lower dose level.
AST or ALT > 3 × ULN with
total bilirubin > 2 × ULN in the
absence of alternative causes
• Permanently discontinue
KRAZATI.
Reference ID: 5095255
3
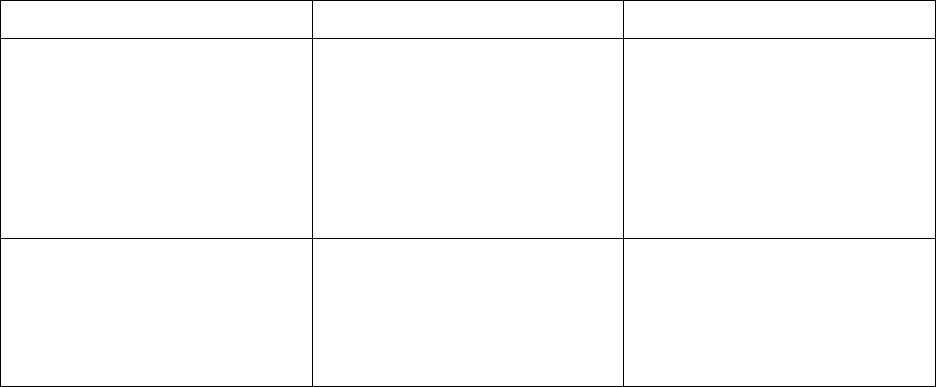
Adverse reaction Severity
a
Dosage Modification
Interstitial Lung Disease /
Pneumonitis
[see Warnings and Precautions
(5.4)]
Any Grade
• Withhold KRAZATI if
ILD/pneumonitis is
suspected.
• Permanently discontinue
KRAZATI if
ILD/pneumonitis is
confirmed.
Other Adverse Reactions
[see Adverse Reactions (6.1)]
Grade 3 or 4
• Withhold KRAZATI until
≤ Grade 1 or return to
baseline.
• Resume KRAZATI at the
next lower dose level.
ALT = alanine aminotransferase; AST = aspartate aminotransferase; ULN = upper limit of normal
a
Grading defined by National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE)
version 5.0
3 DOSAGE FORMS AND STRENGTHS
Tablets: 200 mg, oval shaped, white to off-white, immediate release film coated tablets with
“200” on one side and stylized “M” on the opposite side.
4 CONTRAINDICATIONS
None.
5 WARNINGS AND PRECAUTIONS
5.1 Gastrointestinal Adverse Reactions
KRAZATI can cause severe gastrointestinal adverse reactions.
In the pooled safety population [see Adverse Reactions (6.1)], serious gastrointestinal adverse
reactions observed were gastrointestinal bleeding in 3.8% including 0.8% Grade 3 or 4,
gastrointestinal obstruction in 1.6% including 1.4% Grade 3 or 4, colitis in 0.5% including 0.3%
Grade 3, ileus in 0.5%, and stenosis in 0.3%. In addition, nausea, diarrhea, or vomiting occurred
in 89% of 366 patients, including 9% Grade 3. Nausea, diarrhea, or vomiting led to dosage
interruption or dose reduction in 29% of patients and permanent discontinuation of adagrasib in
0.3%.
Reference ID: 5095255
4
Monitor and manage patients using supportive care, including antidiarrheals, antiemetics, or fluid
replacement, as indicated. Withhold, reduce the dose, or permanently discontinue KRAZATI
based on severity [see Dosage and Administration (2.3)].
5.2 QTc Interval Prolongation
KRAZATI can cause QTc interval prolongation, which can increase the risk for ventricular
tachyarrhythmias (e.g., torsades de pointes) or sudden death.
In the pooled safety population [see Adverse Reactions (6.1)], 6% of 366 patients with at least
one post-baseline electrocardiogram (ECG) assessment had an average QTc ≥ 501 ms and 11%
of patients had an increase from baseline of QTc > 60 msec. KRAZATI causes concentration-
dependent increases in the QTc interval [see Clinical Pharmacology (12.2)].
Avoid concomitant use of KRAZATI with other products with a known potential to prolong the
QTc interval. [see Drug Interactions (7.3) and Clinical Pharmacology (12.2)]. Avoid use of
KRAZATI in patients with congenital long QT syndrome and in patients with concurrent QTc
prolongation.
Monitor ECGs and electrolytes prior to starting KRAZATI, during concomitant use, and as
clinically indicated in patients with congestive heart failure, bradyarrhythmias, electrolyte
abnormalities, and in patients who are unable to avoid concomitant medications that are known
to prolong the QT interval. Withhold, reduce the dose, or permanently discontinue KRAZATI
depending on severity [see Dosage and Administration (2.3)].
5.3 Hepatotoxicity
KRAZATI can cause hepatotoxicity, which may lead to drug-induced liver injury and hepatitis.
In the pooled safety population of 366 patients [see Adverse Reactions (6.1)], drug-induced liver
injury was reported in 0.3% of patients, including 0.3% Grade 3. A total of 32% of patients who
received adagrasib had increased alanine aminotransferase (ALT)/increased aspartate
aminotransferase (AST); 5% were Grade 3 and 0.5% were Grade 4. The median time to first
onset of increased ALT/AST was 3 weeks (range: 0.1 to 48). Overall hepatotoxicity occurred in
37%, and 7% were Grade 3 or 4. Hepatotoxicity leading to dose interruption or reduction
occurred in 12% of patients. Adagrasib was discontinued due to hepatotoxicity in 0.5% of
patients.
Monitor liver laboratory tests (AST, ALT, alkaline phosphatase and total bilirubin) prior to the
start of KRAZATI and monthly for 3 months or as clinically indicated, with more frequent
testing in patients who develop transaminase elevations. Reduce the dose, withhold, or
permanently discontinue KRAZATI based on severity [see Dosage and Administration (2.3) and
Adverse Reactions (6.1)].
Reference ID: 5095255
5
5.4 Interstitial Lung Disease /Pneumonitis
KRAZATI can cause interstitial lung disease (ILD)/pneumonitis, which can be fatal.
In the pooled safety population [see Adverse Reactions (6.1)], ILD/pneumonitis occurred in
4.1% of patients, 1.4% were Grade 3 or 4, and one case was fatal. The median time to first onset
for ILD/pneumonitis was 12 weeks (range: 5 to 31 weeks). Adagrasib was discontinued due to
ILD/pneumonitis in 0.8% of patients.
Monitor patients for new or worsening respiratory symptoms indicative of ILD/pneumonitis
(e.g., dyspnea, cough, fever) during treatment with KRAZATI. Withhold KRAZATI in patients
with suspected ILD/pneumonitis and permanently discontinue KRAZATI if no other potential
causes of ILD/pneumonitis are identified [see Dosage and Administration (2.3)].
6 ADVERSE REACTIONS
The following clinically significant adverse reactions are described elsewhere in the labeling:
• Gastrointestinal Adverse Reactions [see Warnings and Precautions (5.1)]
• QTc Interval Prolongation [see Warnings and Precautions (5.2)]
• Hepatotoxicity [see Warnings and Precautions (5.3)]
• Interstitial Lung Disease (ILD)/Pneumonitis [see Warnings and Precautions (5.4)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates
observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials
of another drug and may not reflect the rates observed in practice.
The pooled safety population described in the WARNINGS AND PRECAUTIONS reflect
exposure to adagrasib as a single agent at 600 mg orally twice daily in 366 patients with NSCLC
and other solid tumors enrolled in KRYSTAL-1 and KRYSTAL-12 (NCT04685135). Among
366 patients who received adagrasib, 39% of patients were exposed for 6 months or longer and
12% were exposed for greater than one year. In this pooled safety population the most common
(≥ 25%) adverse reactions were nausea (70%), diarrhea (69%), vomiting (57%), fatigue (55%),
musculoskeletal pain (38%), hepatotoxicity (37%), renal impairment (33%), edema (30%),
dyspnea (26%), and decreased appetite (29%). In this pooled safety population, the most
common Grade 3 or 4 (≥ 2%) laboratory abnormalities were decreased lymphocytes (20%),
decreased hemoglobin (7%), increased alanine aminotransferase (4.5%), increased aspartate
aminotransferase (4.2%), hypokalemia (3.6%), hyponatremia (3.4%), increased lipase (2.5%),
decreased leukocytes (2.5%), decreased neutrophils (2.3%), and increased alkaline phosphatase
(2.0%).
Reference ID: 5095255
6

Non-Small Cell Lung Cancer
The safety of adagrasib was evaluated in patients with KRAS G12C-mutated, locally advanced or
metastatic NSCLC in KRYSTAL-1 [see Clinical Studies (14)]. Patients received adagrasib
600 mg orally twice daily (n = 116). Among patients who received adagrasib, 45% were exposed
for 6 months or longer and 4% were exposed for greater than one year.
The median age of patients who received adagrasib was 64 years (range 25 to 89), 56% female,
84% White, 8% Black or African American, and 4.3% Asian.
Serious adverse reactions occurred in 57% of patients who received adagrasib. Serious adverse
reactions in ≥ 2% of patients were pneumonia (17%), dyspnea (9%), renal impairment (8%),
sepsis (5%), hypoxia (4.3%), pleural effusion (4.3%), respiratory failure (4.3%), anemia (3.4%),
cardiac failure (3.4%), hyponatremia (3.4%), hypotension (3.4%), muscular weakness (3.4%),
pyrexia (3.4%), dehydration (2.6%), diarrhea (2.6%), mental status changes (2.6%), pulmonary
embolism (2.6%), and pulmonary hemorrhage (2.6%). Fatal adverse reactions occurred in 11%
of patients who received adagrasib due to pneumonia (3.4%), respiratory failure (1.7%), sudden
death (1.7%), cardiac failure (0.9%), cerebrovascular accident (0.9%), mental status change
(0.9%), pulmonary embolism (0.9%), and pulmonary hemorrhage (0.9%).
Permanent discontinuation of adagrasib due to an adverse reaction occurred in 13% of patients.
Adverse reactions which resulted in permanent discontinuation of adagrasib occurring in two
patients each (1.7%) were pneumonia and pneumonitis and occurring in one patient each (0.9%)
were cerebrovascular accident, dyspnea, decreased ejection fraction, encephalitis, gastrointestinal
obstruction, hemorrhage, hepatotoxicity, hypotension, muscular weakness, pulmonary embolism,
pyrexia, respiratory failure and sepsis.
Dose interruptions of adagrasib due to an adverse reaction occurred in 77% of patients. Adverse
reactions requiring dosage interruption in ≥ 2% of patients who received adagrasib included
nausea, hepatotoxicity, fatigue, vomiting, pneumonia, renal impairment, diarrhea, QTc interval
prolongation, anemia, dyspnea, increased lipase, decreased appetite, dizziness, hyponatremia,
muscular weakness, increased amylase, pneumonitis, sepsis and decreased weight.
Dose reductions of adagrasib due to an adverse reaction occurred in 28% of patients. Adverse
reactions which required dose reductions in ≥ 2% of patients who received adagrasib included
hepatotoxicity, fatigue, nausea, diarrhea, vomiting, and renal impairment.
The most common adverse reactions (≥ 20%) were diarrhea, nausea, fatigue, vomiting,
musculoskeletal pain, hepatotoxicity, renal impairment, dyspnea, edema, decreased appetite,
cough, pneumonia, dizziness, constipation, abdominal pain, and QTc interval prolongation. The
most common laboratory abnormalities (≥ 25%) were decreased lymphocytes, increased
aspartate aminotransferase, decreased sodium, decreased hemoglobin, increased creatinine,
decreased albumin, increased alanine aminotransferase, increased lipase, decreased platelets,
decreased magnesium, and decreased potassium.
Table 3 summarizes the adverse reactions in KRYSTAL-1.
Reference ID: 5095255
7
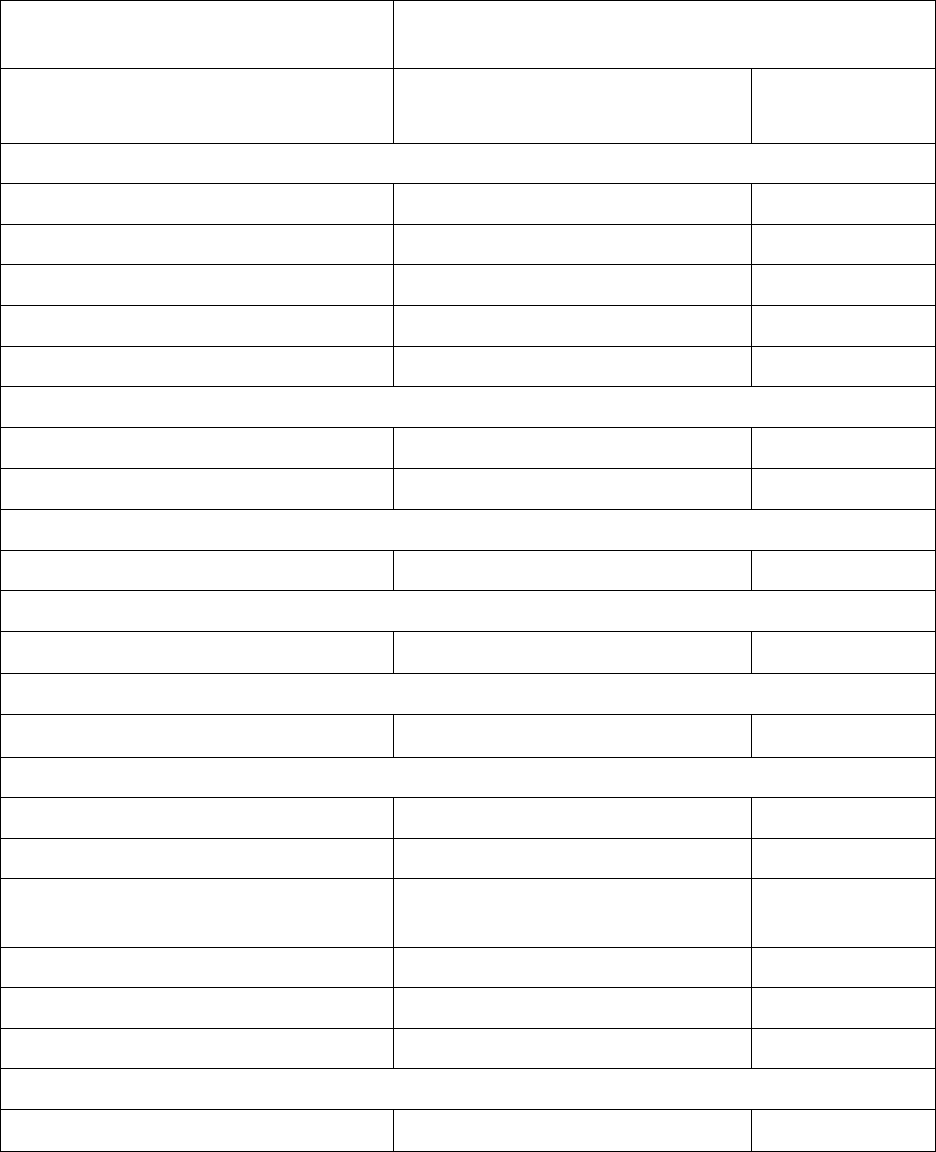
Table 3: Adverse Reactions (≥ 20%) in Patients with KRAS G12C-mutated NSCLC
Who Received Adagrasib in KRYSTAL-1
Adverse Reaction
Adagrasib
N = 116
All Grades
(%)
Grade 3 or 4
(%)
Gastrointestinal Disorders
Diarrhea
*
70 0.9
Nausea 69 4.3
Vomiting
*
56 0.9
Constipation 22 0
Abdominal pain
*
21 0
General Disorders and Administration Site Conditions
Fatigue
*
59 7
Edema
*
32 0
Musculoskeletal and Connective Tissue Disorders
Musculoskeletal pain
*
41 7
Hepatobiliary Disorders
Hepatotoxicity
*,
1
37 10
Renal and Urinary Disorders
Renal impairment
*,
2
36 6
Respiratory
Dyspnea
*
35 10
Cough
*
24 0.9
Metabolism and Nutrition
Disorders
Decreased appetite 30 4.3
Infections and Infestations
Pneumonia
*
24 17
Nervous System Disorders
Dizziness
*
23 0.9
Reference ID: 5095255
8
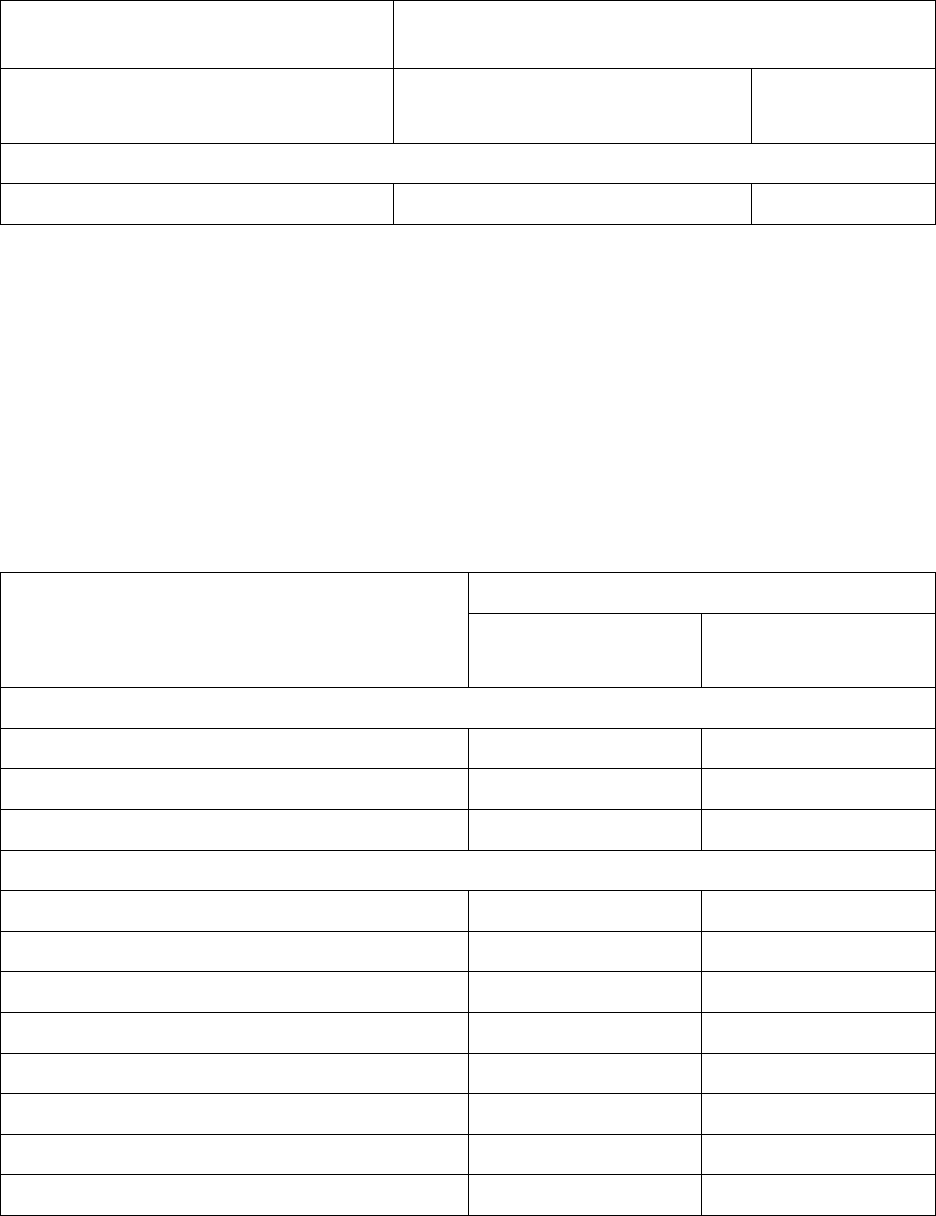
Adverse Reaction
Adagrasib
N = 116
All Grades
(%)
Grade 3 or 4
(%)
Cardiac Disorders
Electrocardiogram QT prolonged 20 6
*Grouped term.
1
Hepatotoxicity includes mixed liver injury, blood alkaline phosphatase increased, alanine aminotransferase
increased, aspartate aminotransferase increased, liver function test increased, blood bilirubin increased, and
bilirubin conjugated increased.
2
Renal impairment includes acute kidney injury and increased blood creatinine.
Table 4 summarizes the laboratory abnormalities in KRYSTAL-1.
Table 4: Select Laboratory Abnormalities Occurring (≥ 25%) That Worsened from
Baseline in Patients with KRAS G12C-mutated NSCLC Who Received
Adagrasib in KRYSTAL-1
Laboratory Abnormality
Adagrasib
*
All Grades
(%)
Grade 3 or 4
(%)
Hematology
Decreased lymphocytes 64 25
Decreased hemoglobin 51 8
Decreased platelets 27 0
Chemistry
Increased aspartate aminotransferase 52 6
Decreased sodium 52 8
Increased creatinine 50 0
Decreased albumin 50 0.9
Increased alanine aminotransferase 46 5
Increased lipase 35 1.8
Decreased magnesium 26 0
Decreased potassium 26 3.5
* Denominator used to calculate the rate varied from 106 to 113 based on the number of patients with a baseline
value and at least one post-treatment value.
Reference ID: 5095255
9

7 DRUG INTERACTIONS
7.1 Effects of Other Drugs on KRAZATI
Strong CYP3A4 Inducers
Avoid concomitant use of KRAZATI with strong CYP3A inducers.
Adagrasib is a CYP3A4 substrate. Concomitant use of KRAZATI with a strong CYP3A inducer
reduces adagrasib exposure [see Clinical Pharmacology (12.3)], which may reduce the
effectiveness of KRAZATI.
Strong CYP3A4 Inhibitors
Avoid concomitant use of KRAZATI with strong CYP3A inhibitors until adagrasib
concentrations have reached steady state (after approximately 8 days).
Adagrasib is a CYP3A4 substrate. If adagrasib concentrations have not reached steady state,
concomitant use of a strong CYP3A inhibitor will increase adagrasib concentrations, [see
Clinical Pharmacology (12.3)], which may increase the risk of KRAZATI adverse reactions.
7.2 Effects of KRAZATI on Other Drugs
Sensitive CYP3A Substrates
Avoid concomitant use of KRAZATI with sensitive CYP3A substrates unless otherwise
recommended in the Prescribing Information for these substrates.
Adagrasib is a CYP3A inhibitor. Concomitant use with KRAZATI increases exposure of
CYP3A substrates [see Clinical Pharmacology (12.3)], which may increase the risk of adverse
reactions related to these substrates.
Sensitive CYP2C9 Substrates
Avoid concomitant use of KRAZATI with sensitive CYP2C9 substrates where minimal
concentration changes may lead to serious adverse reactions unless otherwise recommended in
the Prescribing Information for these substrates.
Adagrasib is a CYP2C9 inhibitor. Concomitant use with KRAZATI increases exposure of
CYP2C9 substrates [see Clinical Pharmacology (12.3)], which may increase the risk of adverse
reactions related to these substrates.
Sensitive CYP2D6 Substrates
Avoid concomitant use of KRAZATI with sensitive CYP2D6 substrates where minimal
concentration changes may lead to serious adverse reactions unless otherwise recommended in
the Prescribing Information for these substrates.
Adagrasib is a CYP2D6 inhibitor. Concomitant use with KRAZATI increases exposure of
CYP2D6 substrates [see Clinical Pharmacology (12.3)], which may increase the risk of adverse
reactions related to these substrates.
Reference ID: 5095255
10

P-gp Substrates
Avoid concomitant use of KRAZATI with P-gp substrates where minimal concentration changes
may lead to serious adverse reactions unless otherwise recommended in the Prescribing
Information for these substrates.
Adagrasib is a P-gp inhibitor. Concomitant use with KRAZATI increases exposure of P-gp
substrates [see Clinical Pharmacology (12.3)], which may increase the risk of adverse reactions
related to these substrates.
7.3 Drugs That Prolong QTc Interval
Avoid concomitant use of KRAZATI with other product(s) with a known potential to prolong the
QTc interval. If concomitant use cannot be avoided, monitor electrocardiogram and electrolytes
prior to starting KRAZATI, during concomitant use, and as clinically indicated [see Warnings
and Precautions (5.2)]. Withhold KRAZATI if the QTc interval is > 500 ms or the change from
baseline is > 60 ms [see Dosage and Administration (2.3)].
Adagrasib causes QTc interval prolongation [see Clinical Pharmacology (12.2)]. Concomitant
use of KRAZATI with other products that prolong the QTc interval may result in a greater
increase in the QTc interval and adverse reactions associated with QTc interval prolongation,
including Torsade de pointes, other serious arrythmias, and sudden death [see Warnings and
Precautions (5.2)].
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
There are no available data on the use of KRAZATI in pregnant women. In animal reproduction
studies, oral administration of adagrasib to pregnant rats and rabbits during the period of
organogenesis did not cause adverse development effects or embryo-fetal lethality at exposures
below the human exposure at the recommended dose of 600 mg twice daily (see Data).
In the U.S. general population, the estimated background risk of major birth defects and
miscarriage in clinically recognized pregnancies is 2% to 4% and 15% to 20%, respectively.
Data
Animal Data
In a rat embryo-fetal development study, once daily oral administration of adagrasib to pregnant
rats during the period of organogenesis resulted in maternal toxicity (reduced body weight and
food intake, and adverse clinical signs leading to moribund condition and early termination) and
lower fetal body weight at 270 mg/kg dose level (approximately 2 times the recommended dose
of 600 mg twice daily based on body surface area [BSA]). Adagrasib induced skeletal
Reference ID: 5095255
11

malformations, such as bent limbs, and skeletal variations, such as bent scapula, wavy ribs, and
supernumerary short cervical ribs at 270 mg/kg, which were secondary to maternal toxicity and
reduced fetal body weight.
In a rabbit embryo-fetal development study, once daily oral administration of adagrasib during
the period of organogenesis resulted in lower fetal body weight and increased litter frequency of
unossified sternebra at 30 mg/kg (approximately 0.11 times the human exposure based on area
under the curve [AUC] at the clinical dose of 600 mg twice daily). This skeletal variation was
associated with maternal toxicities, including reduced mean body weight and decreased food
consumption. Adagrasib exposure did not cause adverse developmental effects and did not affect
embryo-fetal survival in rabbits at doses up to 30 mg/kg once daily.
8.2 Lactation
Risk Summary
There are no data on the presence of adagrasib or its metabolites in human milk, the effects on
the breastfed child, or on milk production. Because of the potential for serious adverse reactions
in breastfed children, advise women not to breastfeed during treatment with KRAZATI and for
1 week after the last dose.
8.3 Females and Males of Reproductive Potential
Infertility
Based on findings from animal studies, KRAZATI may impair fertility in females and males of
reproductive potential [see Nonclinical Toxicology (13.1)].
8.4 Pediatric Use
The safety and effectiveness of KRAZATI has not been established in pediatric patients.
8.5 Geriatric Use
Of 116 patients who received adagrasib 600 mg orally twice daily in KRYSTAL-1, 49%
(57 patients) were ≥ 65 years of age. No overall differences in safety or effectiveness were
observed between older and younger patients.
11 DESCRIPTION
Adagrasib is an irreversible inhibitor of KRAS G12C and belongs to the RAS GTPase family.
The molecular formula is C
32
H
35
ClFN
7
O
2
and the molecular weight is 604.1 g/mol. The
Reference ID: 5095255
12
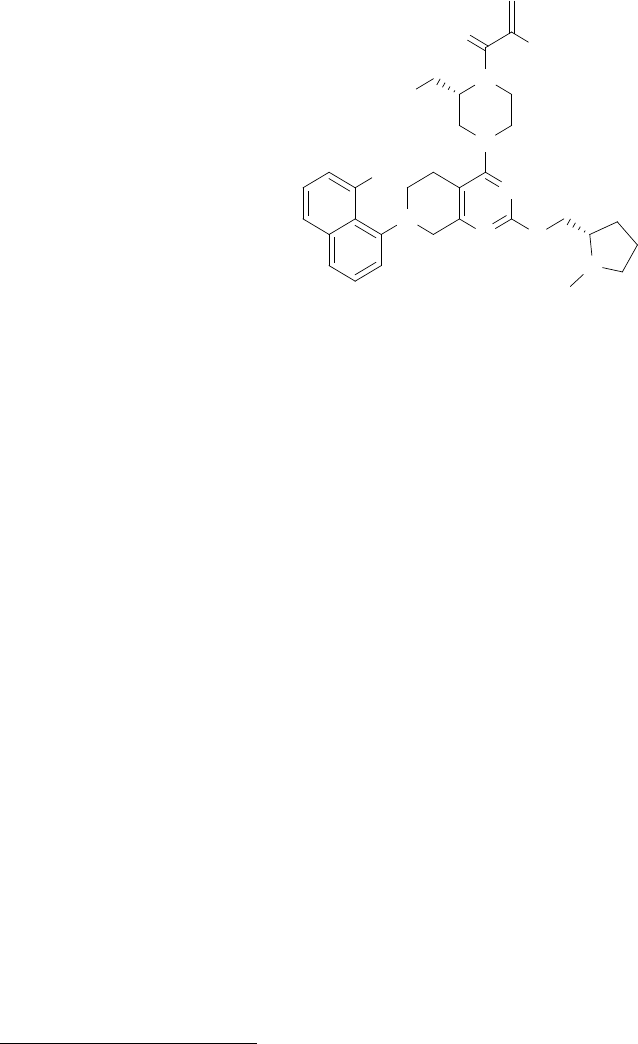
chemical name is {(2S)-4-[7-(8-chloronaphthalen-1-yl)-2-{[(2S)-1-methylpyrrolidin-2-yl]-
methoxy}-5,6,7,8-tetrahydropyrido[3,4-d]pyrimidin-4-yl]-1-(2-fluoroprop-2-enoyl)piperazin-2-
yl}acetonitrile. Adagrasib has the following chemical structure:
O
F
N
NC
N
Cl
N
N
N O
N
Adagrasib is a crystalline solid. The solubility of adagrasib in the aqueous media decreases over
the range pH 1.2 to 7.4 from > 262 mg/mL to < 0.010 mg/mL.
KRAZATI (adagrasib) tablets for oral administration contain 200 mg of adagrasib. The
following are inactive ingredients: colloidal silicon dioxide, crospovidone, magnesium stearate
(vegetable sourced), mannitol, and microcrystalline cellulose. The tablet film coating contains
hypromellose, maltodextrin, medium chain triglycerides (vegetable sourced), polydextrose, talc,
and titanium dioxide.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Adagrasib is an irreversible inhibitor of KRAS G12C that covalently binds to the mutant cysteine
in KRAS G12C and locks the mutant KRAS protein in its inactive state that prevents
downstream signaling without affecting wild-type KRAS protein. Adagrasib inhibits tumor cell
growth and viability in cells harboring KRAS G12C mutations and results in tumor regression in
KRAS G12C-mutated tumor xenograft models with minimal off-target activity.
12.2 Pharmacodynamics
Adagrasib exposure-response relationships and the time course of pharmacodynamic response
are unknown.
Cardiac Electrophysiology
Adagrasib increased QTc in a concentration-dependent manner. Based on the concentration-
QTcF relationship, the mean (90% CI) QTcF change from baseline (ΔQTcF) was 18 (15, 21) ms
at the mean steady-state maximum concentration (C
max,ss
) in patients after administration of
adagrasib 600 mg twice daily [see Warnings and Precautions (5.2)].
Reference ID: 5095255
13

12.3 Pharmacokinetics
The pharmacokinetics of adagrasib were studied in healthy subjects and in patients with KRAS
G12C-mutated NSCLC and are presented as mean (percent coefficient of variation) unless
otherwise specified.
Adagrasib AUC and C
max
increase dose proportionally over the dose range of 400 mg to 600 mg
(0.67 to 1 times the approved recommended dose). Adagrasib steady-state was reached within
8 days following administration of the approved recommended dosage and accumulation was
approximately 6-fold.
Absorption
The median (min, max) T
max
of adagrasib is approximately 6 (6, 12) hours.
Effect of Food
No clinically significant differences in the pharmacokinetics of adagrasib were observed
following administration of a high-fat and high-calorie meal (containing approximately 900 to
1000 calories, 50% from fat).
Distribution
The apparent volume of distribution of adagrasib is 942 L (57%). Human plasma protein binding
of adagrasib is approximately 98% in vitro.
Elimination
The adagrasib terminal elimination half-life is 23 hours (16%) and the apparent oral clearance
(CL/F) is 37 L/h (54%) in patients.
Metabolism
Adagrasib is metabolized primarily by CYP3A4 following single dose administration. Adagrasib
inhibits its own CYP3A4 metabolism following multiple dosing to steady-state which permits
CYP2C8, CYP1A2, CYP2B6, CYP2C9, and CYP2D6 to contribute to its metabolism at steady-
state.
Excretion
Following a single oral dose of radiolabeled adagrasib, approximately 75% of the dose was
recovered in feces (14% as unchanged) and 4.5% recovered in urine (2% as unchanged).
Specific Populations
No clinically significant differences in the pharmacokinetics of adagrasib based on age (19 to
89 years), sex, race (White, Black or African American, or Asian), body weight (36 to 139 kg),
ECOG PS (0, 1), or tumor burden. No clinically significant differences in the pharmacokinetics
of adagrasib are expected in patients with mild to severe renal impairment (CLcr 15 to
< 90 mL/min estimated by Cockcroft-Gault equation) or in patients with mild to severe hepatic
impairment (Child-Pugh classes A to C).
Reference ID: 5095255
14
Drug Interaction Studies
Clinical Studies and Model-Informed Approaches
The following table describes the effect of other drugs on the pharmacokinetics of adagrasib.
Table 5: Effect of Other Drugs on Adagrasib
Concomitant Drug
Adagrasib
Dosage
Changes in C
max
or AUC of
Adagrasib
C
max
% Decrease
AUC
% Decrease
Rifampin
(a strong CYP3A
inducer)
600 mg
single dose
88% 95%
600 mg
multiple doses
> 61%
a
> 66%
a
C
max
= maximum plasma concentration; AUC = area under the plasma concentration-time curve
a
Predicted changes in C
max
or AUC of adagrasib
Strong CYP3A Inhibitors: Adagrasib C
max
increased by 2.4-fold and AUC increased by 4-fold
following concomitant use of a single dose of 200 mg (0.33 times the approved recommended
dose) with itraconazole (a strong CYP3A inhibitor). No clinically significant differences in the
pharmacokinetics of adagrasib at steady state were predicted when used concomitantly with
itraconazole.
No clinically significant differences in the pharmacokinetics of adagrasib were predicted or
observed when used concomitantly with efavirenz (a moderate CYP3A inducer), pantoprazole (a
proton pump inhibitor), or rosuvastatin (a BCRP/OATP substrate).
The following table describes the effect of adagrasib on the pharmacokinetics of other drugs.
Table 6: Effect of Adagrasib on Other Drugs
Concomitant Drug
Adagrasib
Dosage
Fold Increase of Concomitant Drug
C
max
AUC
Midazolam
400 mg
a
twice daily
4.8-fold 21-fold
(a sensitive CYP3A substrate)
600 mg
twice daily
3.1-fold
b
31-fold
b
Reference ID: 5095255
15
Concomitant Drug
Adagrasib
Dosage
Fold Increase of Concomitant Drug
C
max
AUC
Warfarin
(a sensitive CYP2C9
substrate)
600 mg
twice daily
1.1-fold
b
2.9-fold
b
Dextromethorphan
(a sensitive CYP2D6
substrate)
400 mg
a
twice daily
1.9-fold 1.8-fold
600 mg
twice daily
1.7-fold
b
2.4-fold
b
Digoxin
(a P-gp substrate)
600 mg
twice daily
1.9-fold
b
1.5-fold
b
C
max
= maximum plasma concentration; AUC = area under the plasma concentration-time curve
a
0.66 times the approved recommended dosage
b
Predicted changes in C
max
or AUC of concomitant drug
In Vitro Studies
Cytochrome P450 (CYP) Enzymes: Adagrasib may inhibit CYP2B6.
Transporter Systems: Adagrasib may be a substrate of BCRP and may inhibit MATE-1/MATE-
2K.
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with adagrasib.
Adagrasib was not mutagenic in an in vitro bacterial reverse mutation (Ames) assay and was not
genotoxic in an in vitro chromosomal aberration assay or an in vivo micronucleus assay in rats.
Fertility studies were not conducted with adagrasib. In toxicology studies of up to 13-weeks in
duration in rats, oral administration of adagrasib induced phospholipidosis which increased
vacuolation in female reproductive organs, including vacuolation in ovaries (corpora lutea,
macrophage or interstitial cells) and uterus (glandular epithelium), and atrophy with mucification
of the vaginal mucosa at doses ≥ 150 mg/kg (approximately equal to or greater than the human
exposure at the recommended dose based on area under the curve [AUC]). These findings
reversed after cessation of dosing in the 28-day study but in the 13-week study, pigmented
macrophage aggregates were observed in the ovaries of female rats after the recovery period. In a
28-day repeat-dose toxicology study, oral administration of adagrasib to male rats induced
atrophy and epithelial vacuolation of the prostate gland and seminal vesicles at 300 mg/kg
Reference ID: 5095255
16
(approximately 1.6 times the human exposure at the recommended dose based on AUC). These
findings resolved after cessation of treatment.
13.2 Animal Toxicology and/or Pharmacology
Phospholipidosis (vacuolation and/or presence of foamy macrophages) was observed in multiple
organs (e.g., lung, trachea, heart, skeletal, ovaries, uterus, adrenal gland, kidney, liver, lymph
nodes, spleen, thymus, and thyroid in rats; and heart and lung in dogs) after repeated oral
administration of adagrasib in rats and dogs. In toxicology studies of up to 13-week duration in
rats, phospholipidosis was observed at doses ≥ 150 mg/kg (approximately ≥ 2 times the human
exposure at the recommended dose based on AUC). In a dog 28-day toxicity study, this effect
was observed at 25 mg/kg (approximately equal to the human exposure at the recommended dose
based on AUC). The extent of vacuolization and the presence of foamy macrophages were more
prominent in the rat compared to dogs, and evidence of reversibility after cessation of treatment
was noted for most organs. The significance of this finding in humans in unknown.
14 CLINICAL STUDIES
The efficacy of adagrasib was evaluated in KRYSTAL-1 (NCT03785249), a multicenter, single-
arm, open-label expansion cohort study. Eligible patients were required to have locally advanced
or metastatic KRAS G12C-mutated NSCLC who previously received treatment with a platinum-
based regimen and an immune checkpoint inhibitor, an Eastern Cooperative Oncology Group
Performance Status (ECOG PS) of 0 or 1, and at least one measurable lesion as defined by
Response Evaluation criteria in Solid Tumors (RECIST v1.1). Identification of a KRAS G12C
mutation was prospectively determined by local testing using tissue specimens. Patients received
adagrasib 600 mg orally twice daily until unacceptable toxicity or disease progression. Tumor
assessments were performed every 6 weeks. The major efficacy outcome measures were
confirmed objective response rate (ORR) and duration of response (DOR) as evaluated by
blinded independent central review (BICR) according to RECIST v1.1.
In the efficacy population, KRAS G12C mutation status was determined by prospective local
testing using tumor tissue specimens. Of the 112 patients with KRAS G12C mutation, tissue
samples from 88% (98/112) patients were tested retrospectively using the QIAGEN therascreen
KRAS RGQ PCR Kit. While 89% (87/98) of patients were positive for KRAS G12C mutation,
11% (11/98) did not have a KRAS G12C mutation identified. In addition, plasma samples from
63% (71/112) patients were tested retrospectively using Agilent Resolution ctDx FIRST assay.
While 66% (47/71) of patients were positive for KRAS G12C mutation, 34% (24/71) did not have
a KRAS G12C mutation identified.
A total of 112 patients had at least one measurable lesion at baseline as assessed by BICR
according to RECIST v1.1.
The baseline demographic and disease characteristics in the efficacy population were: median
age 64 years (range: 25 to 89), 55% female, 83% White, 8% were Black or African American,
Reference ID: 5095255
17

4% Asian, 4% race not reported, 0.9% American Indian or Alaska Native, 16% Eastern
Cooperative Oncology Group (ECOG) performance status (PS) 0 and 83% ECOG PS 1. Tumor
histology was 97% adenocarcinoma and 89% of patients had metastatic disease. Patients
received a median of 2 prior systemic therapies (range 1 to 7); 43% received 1 prior line, 35%
received 2 prior lines, 10% received 3 prior lines and 12% received 4 or more prior lines, 98%
received both prior platinum and prior anti-PD-1/PD-L1 therapy. Sites of extra-thoracic disease
included bone 42%, brain 30%, adrenals 21%, and liver 21%.
Efficacy results are summarized in Table 7.
Table 7: Efficacy Results for KRYSTAL-1
Efficacy Parameter
Adagrasib
(n = 112)
Objective Response Rate (95% CI)
a
43 (34, 53)
Complete response rate, % 0.9
Partial response rate, % 42
Duration of Response
a
Median
b
in months (95% CI) 8.5 (6.2, 13.8)
Patients with duration ≥ 6 months
c
, % 58
CI = Confidence Interval
a
Assessed by BICR
b
Estimate using Kaplan-Meier method
c
Observed proportion of patients with duration of response beyond landmark time
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
KRAZATI (adagrasib) tablets, 200 mg, oval shaped, white to off-white, immediate release, film
coated tablets with “200” on one side and stylized “M” on the other side.
KRAZATI (adagrasib) tablets are packaged in high-density polyethylene, white opaque, square
bottles with desiccant and polypropylene, white, child resistant closures with a tamper-proof heat
induction seal.
NDC 80739-812-12: 200 mg, bottle containing 120 tablets.
NDC 80739-812-18: 200 mg, bottle containing 180 tablets.
Storage and Handling
Store tablets at room temperature, 20°C to 25°C (68°F to 77°F). Temperature excursions
between 15°C and 30°C (59°F to 86°F) are permitted [see USP Controlled Room Temperature].
Reference ID: 5095255
18

17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Gastrointestinal Adverse Reactions
Advise patients that KRAZATI can cause severe gastrointestinal adverse reactions and to contact
their healthcare provider for signs or symptoms of severe or persistent gastrointestinal adverse
reactions [see Warnings and Precautions (5.1)].
QTc Interval Prolongation
Advise patients that KRAZATI can cause QTc interval prolongation and to contact their
healthcare provider for signs or symptoms of arrhythmias [see Warnings and Precautions (5.2)].
Hepatotoxicity
Advise patients that KRAZATI can cause hepatotoxicity and to immediately contact their
healthcare provider for signs or symptoms of liver dysfunction [see Warnings and Precautions
(5.3)].
Interstitial Lung Disease (ILD)/Pneumonitis
Advise patients that KRAZATI can cause ILD / pneumonitis and to contact their healthcare
provider immediately for new or worsening respiratory symptoms [see Warnings and
Precautions (5.4)].
Drug Interactions
Advise patients to inform their healthcare providers of all concomitant medications, including
prescription medicines, over-the-counter drugs, vitamins, and herbal products [see Drug
Interactions (7.1)].
Missed Dose
If a dose of KRAZATI is missed by greater than 4 hours, resume dosing at the next scheduled
time [see Dosage and Administration (2.2)].
Lactation
Advise women not to breastfeed during treatment with KRAZATI and for 1 week after the last
dose [see Use in Specific Populations (8.2)].
Infertility
Inform patients that KRAZATI may cause infertility [see Use in Specific Populations (8.3)]
KRAZATI (adagrasib)
Reference ID: 5095255
19
Manufactured for:
Mirati Therapeutics, Inc.
3545 Cray Court
San Diego, CA 92121, U.S.A.
© 2022 Mirati Therapeutics, Inc. All Rights Reserved
Reference ID: 5095255
20

PATIENT INFORMATION
KRAZATI
™
(krah zah tee)
(adagrasib)
tablets
What is KRAZATI?
KRAZATI is a prescription medicine used to treat adults with non-small cell lung cancer (NSCLC):
• that has spread to other parts of the body or cannot be removed by surgery, and
• whose tumor has an abnormal KRAS G12C gene, and
• who have received at least one prior treatment for their cancer.
Your healthcare provider will perform a test to make sure that KRAZATI is right for you.
It is not known if KRAZATI is safe and effective in children.
Before taking KRAZATI, tell your healthcare provider about all of your medical conditions, including if you:
• have any heart problems, including heart failure and congenital long QT syndrome.
• have liver problems.
• are pregnant or plan to become pregnant. It is not known if KRAZATI can harm your unborn baby.
• are breastfeeding or plan to breastfeed. It is not known if KRAZATI passes into your breastmilk. Do not breast feed
during treatment and for 1 week after your last dose of KRAZATI.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter
medicines, vitamins, and herbal supplements. KRAZATI can affect the way other medicines work, and other medicines
can affect how KRAZATI works.
How should I take KRAZATI?
• Take KRAZATI exactly as your healthcare provider tells you to take it. Do not change your dose or stop taking
KRAZATI unless your healthcare provider tells you to.
• Your healthcare provider may change your dose, or temporarily or permanently stop treatment with KRAZATI if you
develop certain side effects.
• Take your prescribed dose of KRAZATI 2 times each day, at about the same time each day.
• Take KRAZATI either with food or without food.
• Swallow KRAZATI tablets whole. Do not chew, crush or split tablets.
• If you vomit after taking a dose of KRAZATI, do not take an extra dose. Take your next dose at your next scheduled
time.
If you miss a dose of KRAZATI, take the dose as soon as you remember. If it has been more than 4 hours, do not take
the dose. Take your next dose of KRAZATI at your next scheduled time. Do not take 2 doses at the same time to make
up for a missed dose.
What are possible side effects of KRAZATI?
KRAZATI can cause serious side effects, including:
• Stomach and intestinal (gastrointestinal) problems. Stomach and intestinal side effects including nausea,
diarrhea, or vomiting, are common with KRAZATI but can also sometimes be severe. KRAZATI can also cause
serious stomach and intestinal side effects such as bleeding, obstruction, inflammation of the colon (colitis), and
narrowing (stenosis).
o Call your healthcare provider if you develop any of the signs or symptoms of stomach or intestinal
problems listed above during treatment with KRAZATI.
o Your healthcare provider may prescribe an antidiarrheal medicine or anti-nausea medicine, or other treatment,
as needed.
• Changes in the electrical activity of your heart called QTc prolongation. Certain changes can occur in the
electrical activity of your heart during treatment with KRAZATI, and can be seen on a test called an
electrocardiogram (ECG or EKG). QTc prolongation can increase your risk for irregular heartbeats that can be life-
threatening, such as torsades de pointes, and can lead to sudden death.
o You should not take KRAZATI if you have congenital long QT syndrome or if you currently have QTc
prolongation. See “Before taking KRAZATI, tell your healthcare provider about all of your medical
conditions, including if you:”
Your healthcare provider should monitor the electrical activity of your heart and the levels of body salts in your
blood (electrolytes) during treatment with KRAZATI if you have heart failure, a slow heart rate, abnormal levels
of electrolytes in your blood, or if you take a medicine that can prolong the QT interval of your heartbeat.
Reference ID: 5095255

Tell your healthcare provider if you feel dizzy, lightheaded, or faint, or if you get abnormal heartbeats
during treatment with KRAZATI.
• Liver problems. Abnormal liver blood test results are common with KRAZATI and can sometimes be severe. Your
healthcare provider should do blood tests before starting and during treatment with KRAZATI to check your liver
function. Tell your healthcare provider right away if you develop any signs or symptoms of liver problems, including:
o
your skin or the white part of your eyes turns
o
nausea or vomiting
yellow (jaundice)
o
dark or “tea-colored” urine
o
bleeding or bruising
o
light-colored stools (bowel movements)
o
loss of appetite
o
tiredness or weakness
o
pain, aching or tenderness on the right side of
your stomach area (abdomen)
• Lung or breathing problems. KRAZATI may cause inflammation of the lungs that can lead to death. Tell your
healthcare provider or get emergency medical help right away if you have new or worsening shortness of breath,
cough, or fever.
The most common side effects of KRAZATI include:
o nausea
o muscle and bone pain
o diarrhea
o kidney problems
o vomiting
o swelling
o tiredness
o breathing trouble
o decreased appetite
Certain abnormal laboratory test results are common with KRAZATI. Your healthcare provider will monitor you for
abnormal laboratory tests, and treat you if needed.
KRAZATI may cause fertility problems in males and females, which may affect your ability to have children. Talk to your
healthcare provider if this is a concern for you.
These are not all of the possible side effects of KRAZATI.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
How should I store KRAZATI?
• Store KRAZATI at room temperature between 68°F to 77°F (20°C to 25°C ).
• KRAZATI comes in a child-resistant container.
• KRAZATI comes with a desiccant (drying agent) in the container to keep the medicine dry. Do not remove the
desiccant from the container after opening. Do not eat or swallow the desiccant.
Keep KRAZATI and all medicines out of the reach of children.
General information about the safe and effective use of KRAZATI.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use
KRAZATI for a condition for which it was not prescribed. Do not give KRAZATI to other people, even if they have the
same symptoms that you have. It may harm them. You can ask your pharmacist or healthcare provider for information
about KRAZATI that is written for health professionals.
What are the ingredients in KRAZATI?
Active ingredient: adagrasib
Inactive ingredients: colloidal silicon dioxide, crospovidone, magnesium stearate (vegetable sourced), mannitol, and
microcrystalline cellulose. The tablet film coating contains hypromellose, maltodextrin, medium chain triglycerides
(vegetable sourced), polydextrose, talc, and titanium dioxide.
Manufactured for: Mirati Therapeutics, Inc. 3545 Cray Court San Diego, CA 92121, U.S.A.
© 2022 Mirati Therapeutics, Inc. All rights reserved
For more information, go to www.KRAZATI.com or call 1-844-MIRATI-1 (1-844-647-2841)
This Patient Information has been approved by the U.S. Food and Drug Administration. Issued: 12/2022
Reference ID: 5095255
