
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use
ELAHERE safely and effectively. See full prescribing information for
ELAHERE.
ELAHERE
TM
(mirvetuximab soravtansine-gynx) injection, for
intravenous use
Initial U.S. Approval: 2022
WARNING: OCULAR TOXICITY
See full prescribing information for complete boxed warning.
• ELAHERE can cause severe ocular toxicities, including visual
impairment, keratopathy, dry eye, photophobia, eye pain, and
uveitis. (5.1, 6.1)
• Conduct an ophthalmic exam including visual acuity and slit
lamp exam prior to initiation of ELAHERE, every other cycle
for the first 8 cycles, and as clinically indicated. (2.3)
• Administer prophylactic artificial tears and ophthalmic topical
steroids. (2.3, 5.1)
• Withhold ELAHERE for ocular toxicities until improvement
and resume at the same or reduced dose. (2.4, 5.1)
• Discontinue ELAHERE for Grade 4 ocular toxicities. (2.4, 5.1)
-----------------------------INDICATIONS AND USAGE--------------------------
ELAHERE is a folate receptor alpha (FRα)-directed antibody and microtubule
inhibitor conjugate indicated for the treatment of adult patients with FRα
positive, platinum-resistant epithelial ovarian, fallopian tube, or primary
peritoneal cancer, who have received one to three prior systemic treatment
regimens. Select patients for therapy based on an FDA-approved test. (1, 2.1)
This indication is approved under accelerated approval based on tumor
response rate and durability of response. Continued approval for this
indication may be contingent upon verification and description of clinical
benefit in a confirmatory trial. (1, 14)
------------------------DOSAGE AND ADMINISTRATION----------------------
• Administer ELAHERE as an intravenous infusion only after dilution in 5%
Dextrose Injection, USP. ELAHERE is incompatible with normal saline.
(2.5)
• The recommended dose of ELAHERE is 6 mg/kg adjusted ideal body
weight administered as an intravenous infusion every 3 weeks until disease
progression or unacceptable toxicity. (2.2)
• Premedicate with a corticosteroid, antihistamine, and antipyretic. (2.3)
• Premedicate with an antiemetic, ophthalmic topical steroids, and
lubricating eye drops. (2.3, 5.1)
• See full Prescribing Information for preparation and administration
instructions and dose modifications for adverse reactions. (2)
---------------------DOSAGE FORMS AND STRENGTHS----------------------
• Injection: 100 mg/20 mL (5 mg/mL) in a single-dose vial. (3)
-------------------------------CONTRAINDICATIONS------------------------------
• None. (4)
------------------------WARNINGS AND PRECAUTIONS-----------------------
• Pneumonitis: Withhold ELAHERE for persistent or recurrent Grade 2
pneumonitis and consider dose reduction. Permanently discontinue
ELAHERE for Grade 3 or 4 pneumonitis. (2.4, 5.2)
• Peripheral Neuropathy: Monitor patients for new or worsening peripheral
neuropathy. Withhold dosage, dose reduce, or permanently discontinue
ELAHERE based on the severity of peripheral neuropathy. (2.4, 5.3)
• Embryo-Fetal Toxicity: ELAHERE can cause fetal harm. Advise of the
potential risk to a fetus and to use effective contraception. (5.4, 8.1, 8.3)
-------------------------------ADVERSE REACTIONS------------------------------
The most common (≥20 %) adverse reactions, including laboratory
abnormalities, were vision impairment, fatigue, increased aspartate
aminotransferase, nausea, increased alanine aminotransferase, keratopathy,
abdominal pain, decreased lymphocytes, peripheral neuropathy, diarrhea,
decreased albumin, constipation, increased alkaline phosphatase, dry eye,
decreased magnesium, decreased leukocytes, decreased neutrophils, and
decreased hemoglobin. (6.1)
To report SUSPECTED ADVERSE REACTIONS, contact ImmunoGen
at 1-833-486-4646 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
----------------------------DRUG INTERACTIONS---------------------------------
Strong CYP3A4 Inhibitors: Closely monitor for ELAHERE adverse reactions.
(7.1)
--------------------------USE IN SPECIFIC POPULATIONS---------------------
• Lactation: Advise not to breastfeed. (8.2)
• Moderate or severe hepatic impairment: Avoid use. (8.7)
See 17 for PATIENT COUNSELING INFORMATION and Medication
Guide.
Revised: 11/2022
FULL PRESCRIBING INFORMATION: CONTENTS*
BOXED WARNING
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Patient Selection
2.2 Recommended Dosage
2.3 Premedication and Prophylactic Regimen
2.4 Dosage Modifications
2.5 Instructions for Preparation and Administration
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Ocular Disorders
5.2 Pneumonitis
5.3 Peripheral Neuropathy
5.4 Embryo-Fetal Toxicity
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
7 DRUG INTERACTIONS
7.1 Effects of Other Drugs on ELAHERE
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Renal Impairment
8.7 Hepatic Impairment
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.6 Immunogenicity
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
15 REFERENCES
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
* Sections or subsections omitted from the full prescribing
information are not listed.
Reference ID: 5077370

FULL PRESCRIBING INFORMATION
WARNING: OCULAR TOXICITY
• ELAHERE can cause severe ocular toxicities, including visual impairment, keratopathy, dry
eye, photophobia, eye pain, and uveitis [see Warnings and Precautions (5.1) and Adverse
Reactions (6.1)].
• Conduct an ophthalmic exam including visual acuity and slit lamp exam prior to initiation of
ELAHERE, every other cycle for the first 8 cycles, and as clinically indicated [see Dosage
and Administration (2.3)].
• Administer prophylactic artificial tears and ophthalmic topical steroids [see Dosage and
Administration (2.3) and Warnings and Precautions (5.1)].
• Withhold ELAHERE for ocular toxicities until improvement and resume at the same or
reduced dose [see Dosage and Administration (2.4) and Warnings and Precautions (5.1)].
• Discontinue ELAHERE for Grade 4 ocular toxicities [see Dosage and Administration (2.4)
and Warnings and Precautions (5.1)].
1 INDICATIONS AND USAGE
ELAHERE
™
is indicated for the treatment of adult patients with folate receptor-alpha (FRα) positive, platinum-
resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer, who have received one to three prior
systemic treatment regimens. Select patients for therapy based on an FDA-approved test [see Dosage and
Administration (2.1)].
This indication is approved under accelerated approval based on tumor response rate and durability of response
[see Clinical Studies (14)]. Continued approval for this indication may be contingent upon verification and
description of clinical benefit in a confirmatory trial.
2 DOSAGE AND ADMINISTRATION
2.1 Patient Selection
Select patients for the treatment of platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal
cancer with ELAHERE based on the presence of FRα tumor expression [see Indications & Usage (1) and
Clinical Studies (14)] using an FDA-approved test.
Information on FDA-approved tests for the measurement of FRα tumor expression is available at
http://www.fda.gov/CompanionDiagnostics.
2.2 Recommended Dosage
The recommended dose of ELAHERE is 6 mg/kg adjusted ideal body weight (AIBW) administered once every
3 weeks (21-day cycle) as an intravenous infusion until disease progression or unacceptable toxicity [see
Dosage and Administration (2.5)].
The total dose of ELAHERE is calculated based on each patient’s AIBW using the following formula:
AIBW = Ideal Body Weight (IBW [kg]) + 0.4*(Actual weight [kg] – IBW)
Female IBW (kg) = 0.9*height(cm) – 92
Reference ID: 5077370

2.3 Premedication and Required Eye Care
Premedication
Administer the premedications in Table 1 prior to each infusion of ELAHERE to reduce the incidence and
severity of infusion related reactions (IRRs), nausea, and vomiting.
Table 1: Premedication Prior to Each ELAHERE Infusion
Premedication Route of Administration Examples (or equivalent)
Administration Time
Prior to ELAHERE
Infusion
Corticosteroid intravenous dexamethasone 10 mg
At least 30 minutes prior
Antihistamine oral or intravenous
diphenhydramine 25 mg to
50 mg
Antipyretic oral or intravenous
acetaminophen 325 mg to
650 mg
Antiemetic oral or intravenous
5-HT
3
serotonin receptor
antagonist or appropriate
alternatives
Before each dose and
thereafter as needed
Consider additional premedications including corticosteroids the day prior to ELAHERE administration for
patients who experienced IRRs.
Ophthalmic Exams and Premedication
Ophthalmic exam: Conduct an ophthalmic exam including visual acuity and slit lamp exam prior to initiation of
ELAHERE, every other cycle for the first 8 cycles, and as clinically indicated.
Ophthalmic Topical Steroids: The use of ophthalmic topical steroids is recommended. The initial prescription
and renewals of any corticosteroid medication should be made only after examination with a slit lamp.
Administer one drop of ophthalmic topical steroids in each eye 6 times daily starting the day prior to each
infusion until day 4; then administer one drop in each eye 4 times daily for days 5-8 of each cycle of ELAHERE
[see Warnings and Precautions (5.1)].
Lubricating Eye Drops: The use of lubricating eye drops at least four times daily and as needed is recommended
during treatment with ELAHERE. Instruct patients to use lubricating eye drops and advise to wait at least 10
minutes after ophthalmic topical steroid administration before instilling lubricating eye drops [see Warnings
and Precautions (5.1)].
2.4 Dosage Modifications
Table 2 provides dose reductions and modifications for adverse reactions. Adjust the schedule of administration
to maintain a 3-week interval between doses.
Table 2: Dosage Reduction Schedule
ELAHERE Dose Levels
Starting Dose 6 mg/kg AIBW
First Dose Reduction 5 mg/kg AIBW
Second Dose Reduction 4 mg/kg AIBW
*
* Permanently discontinue in patients who cannot tolerate 4 mg/kg AIBW.
Reference ID: 5077370
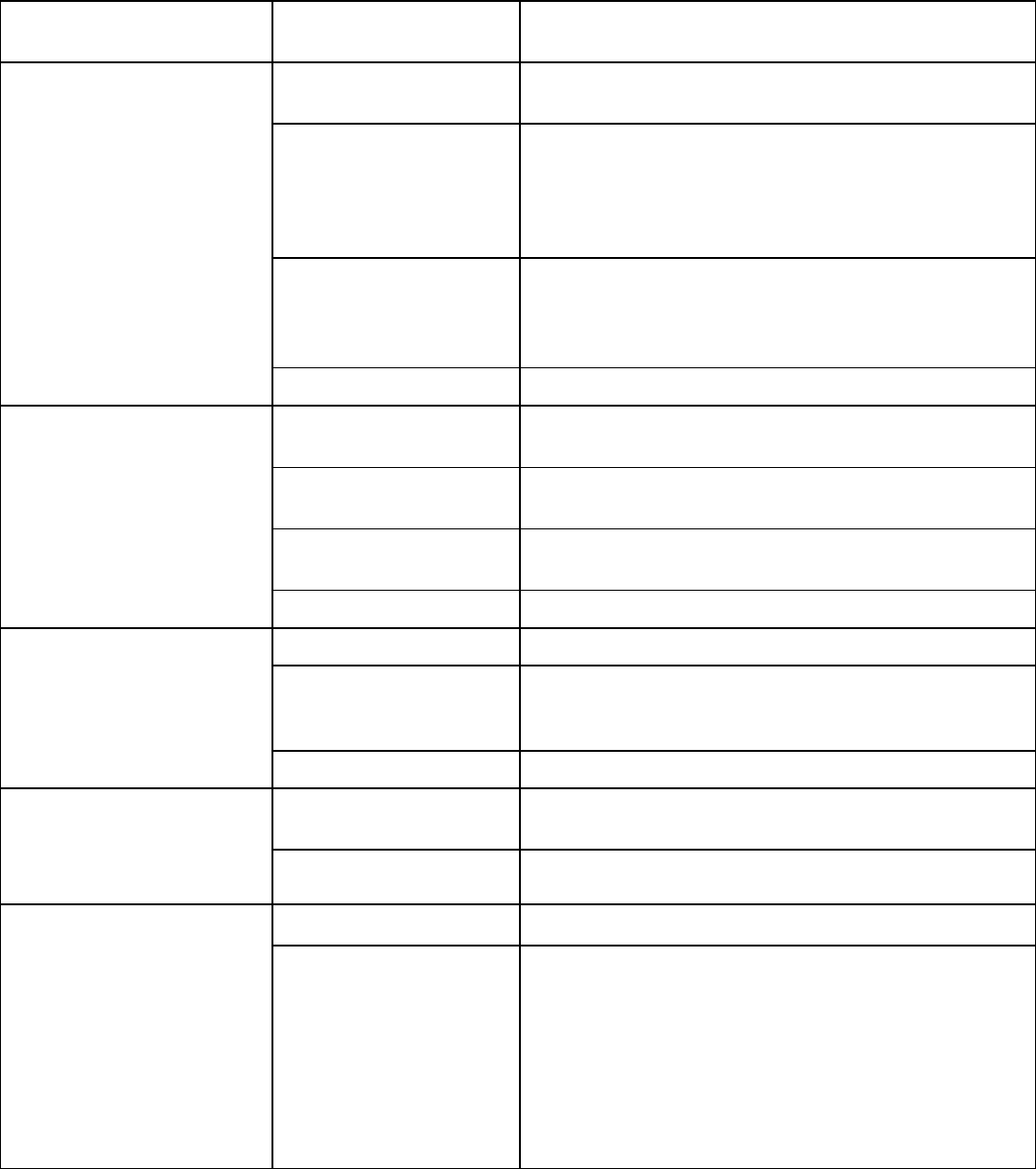
Table 3: Dosage Modifications for Adverse Reactions
Adverse Reaction
Severity of Adverse
Reaction
*
Dosage Modification
Keratitis/Keratopathy
[see Warnings and
Precautions (5.1) and
Adverse Reactions (6.1)]
Nonconfluent superficial
keratitis
Monitor.
Confluent superficial
keratitis, a cornea
epithelial defect, or 3-line
or more loss in best
corrected visual acuity
Withhold dose until improved or resolved, then maintain
at same dose level or consider dose reduction.
Corneal ulcer or stromal
opacity or best corrected
distance visual acuity
20/200 or worse
Withhold dose until improved or resolved, then reduce by
one dose level.
Corneal perforation Permanently discontinue.
Uveitis
[see Warnings and
Precautions (5.1) and
Adverse Reactions (6.1)]
Grade 1/ Rare cell in
anterior chamber
Monitor.
Grade 2/ 1-2+ Cell or
Flare in anterior chamber
Withhold dose until Grade 1 or less, then maintain dose at
same dose level.
Grade 3/ 3+ Cell or Flare
in anterior chamber
Withhold dose until Grade 1 or less, then reduce dose by
one dose level.
Grade 4/ Hypopyon Permanently discontinue.
Pneumonitis
[see Warnings and
Precautions (5.2) and
Adverse Reactions (6.1)]
Grade 1 Monitor.
Grade 2
Withhold dose until Grade 1 or less, then resume at same
dose level or one lower dose level at the discretion of the
healthcare provider.
Grade 3 or 4 Permanently discontinue.
Peripheral Neuropathy
[see Warnings and
Precautions (5.3) and
Adverse Reactions (6.1)]
Grade 2
Withhold dose until Grade 1 or less, then reduce by one
dose level.
Grade 3 or 4 Permanently discontinue.
Infusion-Related
Reactions/Hypersensitivity
[see Adverse Reactions (6.1)]
Grade 1 Maintain infusion rate.
Grade 2
• Interrupt infusion and administer supportive
treatment.
• After recovery from symptoms, resume the infusion at
50% of the previous rate, and if no further symptoms
appear, increase rate as appropriate until infusion is
completed [see Dosage and Administration (2.5)].
• Administer additional premedication for future cycles
[see Dosage and Administration (2.3)].
Reference ID: 5077370

Adverse Reaction
Severity of Adverse
Reaction
*
Dosage Modification
Grade 3 or 4
• Immediately stop infusion and administer supportive
treatment.
• Advise patient to seek emergency treatment and
immediately notify their healthcare provider if the
infusion-related symptoms recur.
• Permanently discontinue.
Other Adverse Reactions
[see Adverse Reactions (6.1)]
Grade 3
Withhold dose until Grade 1 or less, then resume at one
lower dose level.
Grade 4 Permanently discontinue.
*
Unless otherwise specified, National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) version 5.0.
2.5 Instructions for Preparation and Administration
Preparation
• ELAHERE is a hazardous drug. Follow applicable special handling and disposal procedures
1
.
• Calculate the dose (mg) (based on the patient’s AIBW), total volume (mL) of solution required, and
the number of vials of ELAHERE needed [see Recommended Dosage (2.2) and Dose Modifications
(2.4)]. More than one vial will be needed for a full dose.
• Remove the vials of ELAHERE from the refrigerator and allow to warm to room temperature.
• Parenteral drug products should be inspected visually for particulate matter and discoloration prior to
administration, whenever solution and container permit. ELAHERE is a clear to slightly opalescent,
colorless solution.
• Gently swirl and inspect each vial prior to withdrawing the calculated dose volume of ELAHERE
for subsequent further dilution. Do not shake the vial.
• Using aseptic technique, withdraw the calculated dose volume of ELAHERE for subsequent further
dilution.
• ELAHERE contains no preservatives and is intended for single-dose only. Discard any unused drug
remaining in the vial.
Dilution
• ELAHERE must be diluted prior to administration with 5% Dextrose Injection, USP to a final
concentration of 1 mg/mL to 2 mg/mL.
• ELAHERE is incompatible with 0.9% Sodium Chloride Injection. ELAHERE must not be mixed
with any other drugs or intravenous fluids.
• Determine the volume of 5% Dextrose Injection, USP required to achieve the final diluted drug
concentration. Either remove excess 5% Dextrose Injection, USP from a prefilled intravenous bag or
add the calculated volume of 5% Dextrose Injection, USP to a sterile empty intravenous bag. Then
add the calculated dose volume of ELAHERE to the intravenous bag.
• Gently mix the diluted drug solution by slowly inverting the bag several times to assure uniform
mixing. Do not shake or agitate.
• If the diluted infusion solution is not used immediately, store solution either at ambient temperature
[(18°C to 25°C (64.4°F to 77°F)] for no more than 8 hours (including infusion time), or under
Reference ID: 5077370

refrigeration at 2°C to 8°C (36°F to 46°F) for no more than 12 hours. If refrigerated, allow the
infusion bag to reach room temperature prior to administration. After refrigeration, administer
diluted infusion solutions within 8 hours (including infusion time).
• Do not freeze prepared infusion solution.
Administration
• Inspect the ELAHERE intravenous infusion bag visually for particulate matter and discoloration
prior to administration.
• Administer pre-medications prior to ELAHERE administration [see Premedication and Prophylactic
Regimen (2.3)].
• Administer ELAHERE as an intravenous infusion only, using a 0.2 or 0.22 µm polyethersulfone
(PES) in-line filter. Do not substitute other membrane materials.
• Administer the initial dose as an intravenous infusion at the rate of 1 mg/min. If well tolerated after
30 minutes at 1 mg/min, the infusion rate can be increased to 3 mg/min. If well tolerated after 30
minutes at 3 mg/min, the infusion rate can be increased to 5 mg/min.
• If no infusion-related reactions occur with the previous dose, subsequent infusions should be started
at the maximally tolerated rate and may be increased up to a maximum infusion rate of 5 mg/min, as
tolerated.
• Following the infusion, flush the intravenous line with 5% Dextrose Injection, USP to ensure
delivery of the full dose. Do not use any other intravenous fluids for flushing.
3 DOSAGE FORMS AND STRENGTHS
Injection: 100 mg/20 mL (5 mg/mL) clear to slightly opalescent, colorless solution in a single-dose vial.
4 CONTRAINDICATIONS
None.
5 WARNINGS AND PRECAUTIONS
5.1 Ocular Disorders
ELAHERE can cause severe ocular adverse reactions, including visual impairment, keratopathy (corneal
disorders), dry eye, photophobia, eye pain, and uveitis.
Ocular adverse reactions occurred in 61% of patients with ovarian cancer treated with ELAHERE. Nine percent
(9%) of patients experienced Grade 3 ocular adverse reactions, including visual impairment,
keratopathy/keratitis (corneal disorders), dry eye, photophobia, and eye pain; and one patient (0.2%)
experienced Grade 4 keratopathy. The most common (≥5%) ocular adverse reactions were visual impairment
(49%), keratopathy (36%), dry eye (26%), cataract (15%), photophobia (13%), and eye pain (12%) [see Adverse
Reactions (6.1)].
The median time to onset for first ocular adverse reaction was 1.2 months (range: 0.03 to 12.9). Of the patients
who experienced ocular events, 49% had complete resolution and 39% had partial improvement (defined as a
decrease in severity by one or more grades from the worst grade) at last follow up. Ocular adverse reactions led
to permanent discontinuation of ELAHERE in 0.6% of patients.
Premedication and use of lubricating and ophthalmic topical steroid eye drops during treatment with ELAHERE
are recommended [see Dosage and Administration (2.3)]. Advise patients to avoid use of contact lenses during
treatment with ELAHERE unless directed by a healthcare provider.
Reference ID: 5077370
Refer patients to an eye care professional for an ophthalmic exam including visual acuity and slit lamp exam
prior to treatment initiation, every other cycle for the first 8 cycles, and as clinically indicated. Promptly refer
patients to an eye care professional for any new or worsening ocular signs and symptoms.
Monitor for ocular toxicity and withhold, reduce, or permanently discontinue ELAHERE based on severity and
persistence of ocular adverse reactions. [see Dosage and Administration (2.4)].
5.2 Pneumonitis
Severe, life-threatening, or fatal interstitial lung disease (ILD), including pneumonitis, can occur in patients
treated with ELAHERE.
Pneumonitis occurred in 10% of patients treated with ELAHERE, including 0.8% with Grade 3 events, and 1
patient (0.2%) with a Grade 4 event. One patient (0.2%) died due to respiratory failure in the setting of
pneumonitis and lung metastases. Pneumonitis resulted in ELAHERE dose reduction in 1%, dose interruptions
in 3%, and permanent discontinuation in 3% of patients.
Monitor patients for pulmonary signs and symptoms of pneumonitis, which may include hypoxia, cough,
dyspnea, or interstitial infiltrates on radiologic exams. Infectious, neoplastic, and other causes for such
symptoms should be excluded through appropriate investigations. Withhold ELAHERE for patients who
develop persistent or recurrent Grade 2 pneumonitis until symptoms resolve to ≤ Grade 1 and consider dose
reduction. Permanently discontinue ELAHERE in all patients with Grade 3 or 4 pneumonitis [see Dosage and
Administration (2.4)]. Patients who are asymptomatic may continue dosing of ELAHERE with close
monitoring.
5.3 Peripheral Neuropathy
Peripheral neuropathy occurred in 36% of patients with ovarian cancer treated with ELAHERE across clinical
trials; 2% of patients experienced Grade 3 peripheral neuropathy. Peripheral neuropathy adverse reactions
included peripheral neuropathy (19%), peripheral sensory neuropathy (9%), paraesthesia (6%), neurotoxicity
(3%), hypoaesthesia (2%), peripheral motor neuropathy (1%), neuralgia (0.4%), polyneuropathy (0.2%) and
oral hypoesthesia (0.2%).
The median time to onset of peripheral neuropathy was 1.3 months (range 0.03 to 29.1). Of the patients who
experienced peripheral neuropathy, 28% had complete resolution and 13% had partial improvement (defined as
a decrease in severity by one or more grades from the worst grade) at last follow up. Peripheral neuropathy led
to discontinuation of ELAHERE in 0.4% of patients.
Monitor patients for signs and symptoms of neuropathy, such as paresthesia, tingling or a burning sensation,
neuropathic pain, muscle weakness, or dysesthesia. For patients experiencing new or worsening peripheral
neuropathy, withhold dosage, dose reduce, or permanently discontinue ELAHERE based on the severity of
peripheral neuropathy [see Dosage and Administration (2.4)].
5.4 Embryo-Fetal Toxicity
Based on its mechanism of action, ELAHERE can cause embryo-fetal harm when administered to a pregnant
woman because it contains a genotoxic compound (DM4) and affects actively dividing cells.
Advise pregnant women of the potential risk to a fetus. Advise females of reproductive potential to use effective
contraception during treatment with ELAHERE and for 7 months after the last dose [see Use in Specific
Populations (8.1, 8.3)].
6 ADVERSE REACTIONS
The following adverse reactions are discussed elsewhere in the labeling:
• Ocular Disorders [see Warnings and Precautions (5.1)].
Reference ID: 5077370

• Pneumonitis [see Warnings and Precautions (5.2)].
• Peripheral Neuropathy [see Warnings and Precautions (5.3)].
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the
clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not
reflect the rates observed in practice.
The pooled safety population described in WARNINGS AND PRECAUTIONS reflect exposure to ELAHERE
in 464 patients with epithelial ovarian, fallopian tube, or primary peritoneal cancer at 6 mg/kg AIBW
administered intravenously once every 3 weeks until disease progression or unacceptable toxicity in Study
0417; Study 0403 (NCT02631876), and Study 0401 (NCT01609556). The median duration of treatment was 4.3
months (range: 0.7 to 30.4).
Epithelial Ovarian, Fallopian Tube, or Primary Peritoneal Cancer
Study 0417
The safety of ELAHERE was evaluated in Study 0417, a single-arm, open-label study in patients (n=106) with
platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer [see Clinical Studies (14)].
Patients received ELAHERE 6 mg/kg AIBW once every 3 weeks until disease progression or unacceptable
toxicity. The median duration of treatment was 4.2 months (range: 0.7 to 13.3).
Serious adverse reactions occurred in 31% of patients. The most common (≥2%) serious adverse reactions were
intestinal obstruction (8%), ascites (4%), infection (3%), and pleural effusion (3%). Fatal adverse reactions
occurred in 2% of patients, including small intestinal obstruction (1%) and pneumonitis (1%).
Permanent discontinuation of ELAHERE due to adverse reactions occurred in 11% of patients. The most
common (≥2%) adverse reactions leading to permanent discontinuation were intestinal obstruction (2%) and
thrombocytopenia (2%). One patient (0.9%) permanently discontinued ELAHERE due to visual impairment
(unilateral decrease to BCVA < 20/200 that resolved to baseline after discontinuation).
Dosage delays of ELAHERE due to an adverse reaction occurred in 39% of patients treated with ELAHERE.
Adverse reactions which required dosage delays in ≥3% of patients included visual impairment (15%),
keratopathy (11%), neutropenia (6%), dry eye (5%), cataracts (3%), and increased gamma-glutamyltransferase
(3%).
Dose reductions of ELAHERE due to an adverse reaction occurred in 20% of patients. Adverse reactions which
required dose reductions in ≥3% of patients included visual impairment (9%) and keratopathy (7%).
The most common (≥20%) adverse reactions, including laboratory abnormalities, were vision impairment,
fatigue, increased aspartate aminotransferase, nausea, increased alanine aminotransferase, keratopathy,
abdominal pain, decreased lymphocytes, peripheral neuropathy, diarrhea, decreased albumin, constipation,
increased alkaline phosphatase, dry eye, decreased magnesium, decreased leukocytes, decreased neutrophils,
and decreased hemoglobin.
Table 4 summarizes the adverse reactions (≥10%) in patients treated with ELAHERE in Study 0417.
Reference ID: 5077370
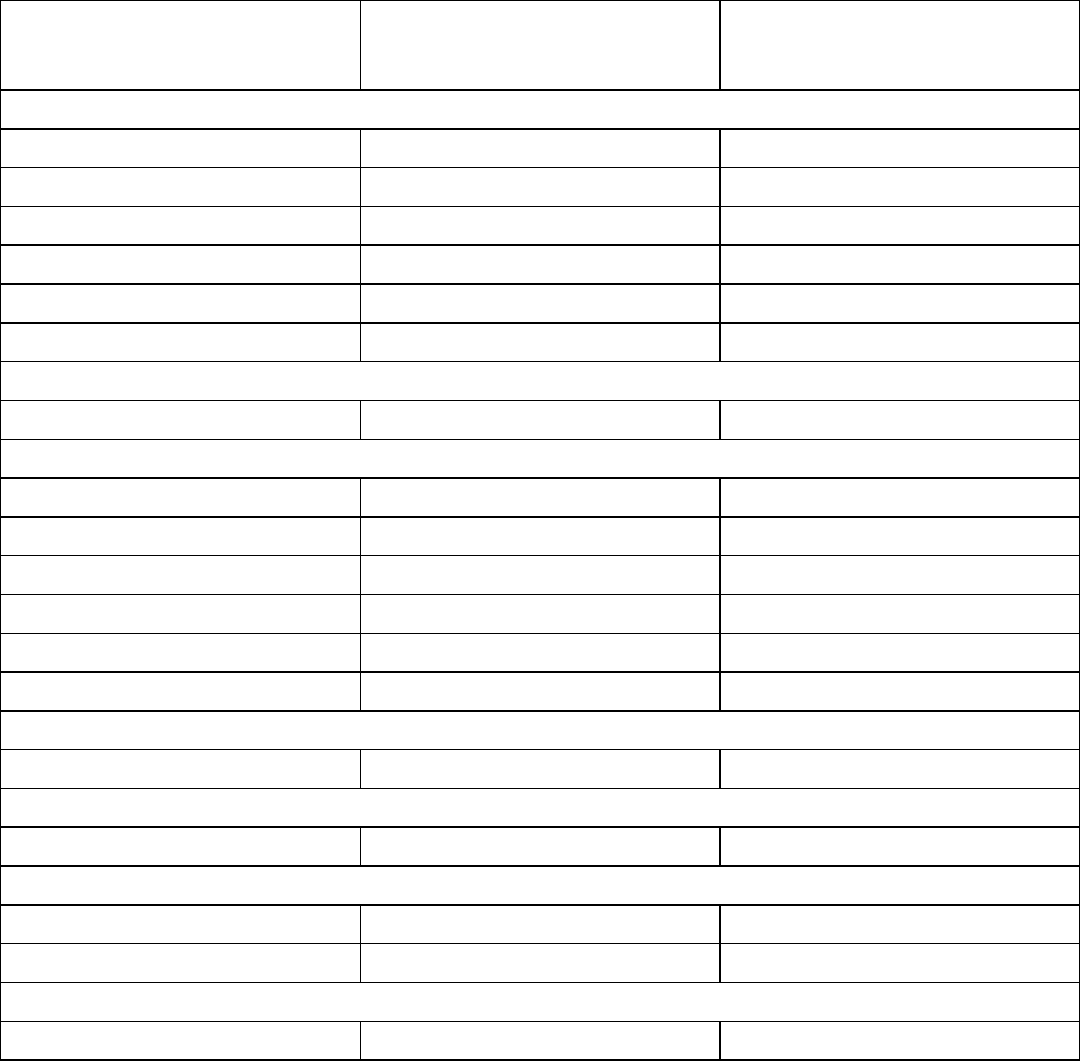
Table 4: Adverse Reactions (≥10%) in Patients with Epithelial Ovarian, Fallopian Tube, or Primary
Peritoneal Cancer Who Received ELAHERE in Study 0417
Adverse Reaction
All Grades
N=106
(%)
Grade 3-4
N=106
(%)
Eye disorders
Vision impairment
※
50 7
Keratopathy
†
37 9
Dry eye
‡
27 2
Cataract 18 3
Photophobia 17 0
Eye Pain
§
10 0
General disorders
Fatigue 49 3
Gastrointestinal disorders
Nausea 40 0
Abdominal Pain
*
36 7
Diarrhea 31 3
Constipation 30 1
Vomiting 19 0
Abdominal distension 11 0
Nervous system disorders
Peripheral neuropathy
¶
33 2
Metabolism and nutrition disorders
Decreased appetite 18 1
Musculoskeletal and connective tissue disorders
Arthralgia 17 0
Myalgia 10 0
Respiratory, thoracic, and mediastinal disorders
Dyspnea^ 12 0
※Visual Impairment includes vision blurred, vitreous floaters, visual acuity reduced, diplopia, presbyopia, accommodation disorder,
visual impairment, and refraction disorder.
† Keratopathy includes corneal disorder, corneal epithelial microcysts, corneal epithelial defect, keratitis, keratopathy, corneal
deposits, and punctate keratitis.
‡ Dry eye includes dry eye and lacrimation increased.
§ Eye pain includes eye pain and ocular discomfort.
⸙
Fatigue includes fatigue and asthenia.
* Abdominal pain includes abdominal pain, abdominal pain upper, abdominal pain lower, abdominal discomfort.
¶
Peripheral neuropathy includes neuropathy peripheral, peripheral sensory neuropathy, peripheral motor neuropathy, paresthesia,
hypoesthesia, polyneuropathy, and neurotoxicity.
^ Dyspnea includes dyspnea and exertional dyspnea.
Reference ID: 5077370
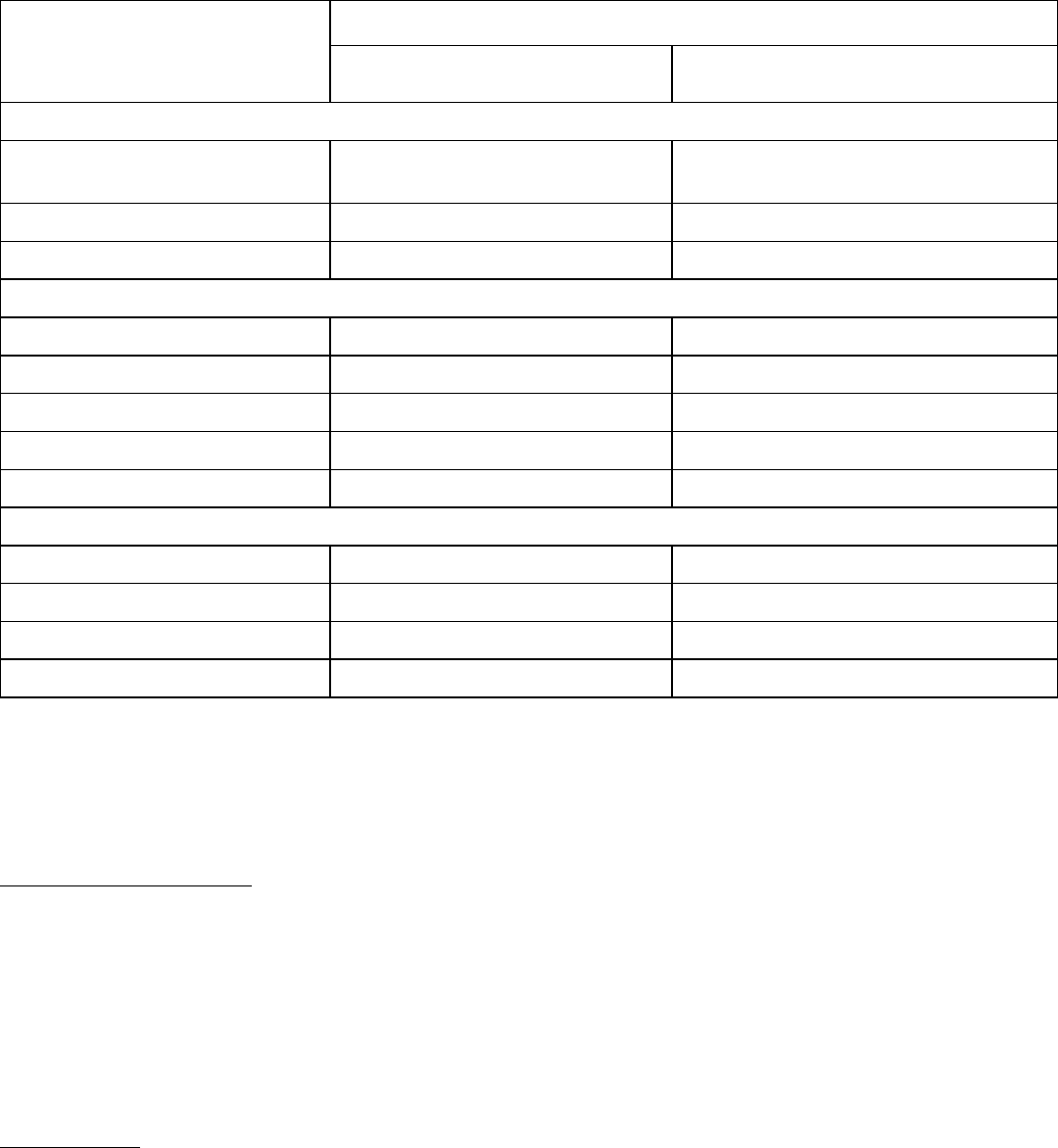
Clinically relevant adverse reactions occurring in <10% of patients who received ELAHERE in Study 0417
included infusion related reactions/hypersensitivity (9%), pneumonitis (8%), thrombocytopenia (5%), and
uveitis (1%).
Table 5 summarizes the laboratory abnormalities in Study 0417.
Table 5: Select Laboratory Abnormalities ≥10% for All Grades, or ≥2% for Grades 3-4 in Patients Who
Received ELAHERE
Laboratory Abnormality
ELAHERE*
All Grades
(%)
Grade 3-4
(%)
Liver Function Tests
Increased aspartate
aminotransferase
50 2
Increased alanine aminotransferase 39 2
Increased alkaline phosphatase 30 1
Hematology*
Decreased lymphocytes 35 7
Decreased leukocytes 26 1
Decreased neutrophils 26 3
Decreased hemoglobin 25 3
Decreased platelets 18 2
Chemistry
Decreased albumin 31 1
Decreased magnesium 27 2
Increased creatinine 16 0
Decreased potassium 15 4
* The denominator used to calculate the rate varied from 98 to 101 based on the number of patients with a baseline value and at least
one post-treatment value.
7 DRUG INTERACTIONS
7.1 Effects of Other Drugs on ELAHERE
Strong CYP3A4 Inhibitors
DM4 is a CYP3A4 substrate. Concomitant use of ELAHERE with strong CYP3A4 inhibitors may increase
unconjugated DM4 exposure [see Clinical Pharmacology (12.3)], which may increase the risk of ELAHERE
adverse reactions [see Adverse Reactions (6)]. Closely monitor patients for adverse reactions with ELAHERE
when used concomitantly with strong CYP3A4 inhibitors [see Warnings and Precautions (5)].
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Risk Summary
Reference ID: 5077370

Based on its mechanism of action, ELAHERE can cause embryo-fetal harm when administered to a pregnant
woman because it contains a genotoxic compound (DM4) and affects actively dividing cells [see Clinical
Pharmacology (12.1), Nonclinical Toxicology (13.1)]. Human immunoglobulin G (IgG) is known to cross the
placental barrier; therefore, ELAHERE has the potential to be transmitted from the mother to the developing
fetus. There are no available human data on ELAHERE use in pregnant women to inform a drug-associated
risk. No reproductive or developmental animal toxicity studies were conducted with mirvetuximab
soravtansine-gynx. Advise patients of the potential risk to a fetus.
The estimated background risk of major birth defects and miscarriage for the indicated population is unknown.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in
clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Data
Animal Data: No reproductive or developmental animal toxicity studies have been conducted with
mirvetuximab soravtansine-gynx. The cytotoxic component of ELAHERE, DM4, disrupts microtubule function,
is genotoxic, and can be toxic to actively dividing cells, suggesting it has the potential to cause embryotoxicity
and teratogenicity.
8.2 Lactation
Risk Summary
There are no data on the presence of mirvetuximab soravtansine-gynx in human milk or the effects on the
breastfed child or milk production. Because of the potential for serious adverse reactions in a breastfed child,
advise women not to breastfeed during treatment with ELAHERE and for 1 month after the last dose.
8.3 Females and Males of Reproductive Potential
ELAHERE can cause embryo-fetal harm when administered to a pregnant woman [see Use in Specific
Populations (8.1)].
Pregnancy Testing
Verify pregnancy status in females of reproductive potential prior to initiating ELAHERE.
Contraception
Females: Advise females of reproductive potential to use effective contraception during treatment with
ELAHERE and for 7 months after the last dose.
8.4 Pediatric Use
Safety and effectiveness of ELAHERE have not been established in pediatric patients.
8.5 Geriatric Use
Of the 106 patients who were treated in Study 0417, 44% of patients were ≥65 years old. Grade ≥3 adverse
reactions occurred in 49% of patients ≥65 years and in 51% <65 years. No clinically meaningful differences in
efficacy or safety were observed between patients ≥65 years of age compared to younger patients.
Population pharmacokinetic analysis indicates that age does not have a clinically meaningful effect on the
pharmacokinetics of ELAHERE [see Clinical Pharmacology (12.3)].
8.6 Renal Impairment
No dosage adjustment of ELAHERE is recommended for patients with mild to moderate renal impairment
(CLcr 30 to 90 mL/min). The effect of severe renal impairment (CLcr 15 to < 30 mL/min) or end-stage renal
disease on ELAHERE is unknown [see Clinical Pharmacology (12.3)].
Reference ID: 5077370
8.7 Hepatic Impairment
Avoid use of ELAHERE in patients with moderate or severe hepatic impairment (total bilirubin >1.5 ULN).
No dosage adjustment of ELAHERE is recommended for patients with mild hepatic impairment (total bilirubin
≤ULN and AST >ULN or total bilirubin >1 to 1.5 times ULN and any AST) [see Clinical Pharmacology
(12.3)].
11 DESCRIPTION
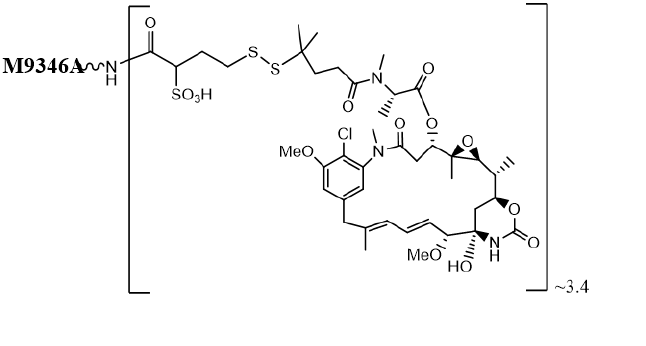
Mirvetuximab soravtansine-gynx is a folate receptor alpha (FRα)-directed antibody-drug conjugate (ADC)
consisting of three components: 1) an anti-FRα monoclonal antibody of IgG1 subtype 2) the small molecule
anti-tubulin agent DM4 (a maytansine derivative) and 3) a linker, sulfo-SPDB (1-(2,5-dioxopyrrolidin-1-yl)oxy-
1-oxo-4-(pyridin-2-yldisulfanyl)butane-2-sulfonic acid) that covalently attaches DM4 to the mirvetuximab
antibody. Mirvetuximab soravtansine-gynx has an approximate molecular weight of 150 kDa. An average of 3.4
molecules of DM4 are attached to each antibody molecule. Mirvetuximab soravtansine-gynx is produced by
chemical conjugation of the antibody and small molecule components. The antibody is produced by mammalian
(Chinese hamster ovary) cells, and the small molecule components are produced by chemical synthesis.
Mirvetuximab soravtansine-gynx has the following structure:
ELAHERE (mirvetuximab soravtansine-gynx) injection is supplied as a sterile, preservative-free, clear to
slightly opalescent, colorless solution containing 100 mg/20 mL of mirvetuximab soravtansine-gynx in single-
dose vials. Each mL of solution contains 5 mg of mirvetuximab soravtansine-gynx, and glacial acetic acid (0.22
mg), polysorbate 20 (0.1 mg), sodium acetate (0.53 mg), sucrose (90 mg), and Water for Injection. The pH is
approximately 5.0.
The ELAHERE vial stoppers are not made with natural rubber latex.
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
Mirvetuximab soravtansine-gynx is an antibody-drug conjugate (ADC). The antibody is a chimeric IgG1
directed against folate receptor alpha (FRα). The small molecule, DM4, is a microtubule inhibitor attached to
the antibody via a cleavable linker. Upon binding to FRα, mirvetuximab soravtansine-gynx is internalized
followed by intracellular release of DM4 via proteolytic cleavage. DM4 disrupts the microtubule network
within the cell, resulting in cell cycle arrest and apoptotic cell death.
Reference ID: 5077370

12.2 Pharmacodynamics
Exposure-Response Relationships
An exposure-response relationship between mirvetuximab soravtansine-gynx and overall response rates was
observed. Higher incidence of Grade ≥2 ocular adverse reactions and Grade ≥2 peripheral neuropathy occurred
with increasing mirvetuximab soravtansine-gynx exposure.
Cardiac Electrophysiology
At the approved recommended dose, ELAHERE did not cause large mean increases (>10 msec) in the QTc
interval.
12.3 Pharmacokinetics
The pharmacokinetics were characterized after patients were administered mirvetuximab soravtansine-gynx
0.161 mg/kg to 8.71 mg/kg adjusted ideal body weight (AIBW) dosages, (0.0268 times to 1.45 times the
approved recommended dosage of 6 mg/kg AIBW), unless otherwise noted.
Table 6 summarizes the exposure parameters of mirvetuximab soravtansine-gynx, unconjugated DM4, and its
metabolite S-methyl-DM4 following administration after the first cycle (3-weeks) of mirvetuximab
soravtansine-gynx 6 mg/kg to patients. Peak mirvetuximab soravtansine-gynx concentrations were observed
near the end of intravenous infusion, while peak unconjugated DM4 concentrations were observed on the
second day after administration of mirvetuximab soravtansine-gynx, and the peak S-methyl-DM4
concentrations were observed approximately 3 days after administration of mirvetuximab soravtansine-gynx.
Steady state concentrations of mirvetuximab soravtansine-gynx, DM4, and S-methyl-DM4 were reached after 1
treatment cycle. Accumulation of the mirvetuximab soravtansine-gynx, DM4, and S-methyl-DM4 was minimal
following repeat administration of mirvetuximab soravtansine-gynx.
Table 6: Exposure Parameters of Mirvetuximab Soravtansine-gynx, Unconjugated DM4, and S-methyl
DM4 After First Treatment Cycle of 6 mg/kg of Mirvetuximab Soravtansine-gynx
Mirvetuximab Soravtansine-gynx
Mean (±SD)
Unconjugated DM4
Mean (±SD)
S-methyl-DM4
Mean (±SD)
C
max
137.3 (±62.3) µg/mL 4.11 (±2.29) ng/mL 6.98 (±6.79) ng/mL
AUC
tau
20.65 (±6.84) h*mg/mL 530 (±245) h*ng/mL 1848 (±1585) h*ng/mL
C
max
= maximum concentration, AUC
tau
= area under the concentration vs. time curve over the dosing interval (21 days).
Distribution
The mean (±SD) steady state volume of distribution of mirvetuximab soravtansine-gynx was 2.63 (±2.98) L.
Human plasma protein binding of DM4 and S-methyl DM4 was >99%, in vitro.
Elimination
Total plasma clearance (geometric mean [CV%]) of mirvetuximab soravtansine-gynx was 18.9 mL/hour
(51.9%). The geometric mean terminal phase half-life of mirvetuximab soravtansine-gynx after the first dose
was 4.8 days leading to a steady state at approximately 24 days. For the unconjugated DM4, the total plasma
clearance (geometric mean [CV%]) was 13.8 L/hour (31.1%) and the geometric mean terminal phase half-life
was 2.8 days. For S-methyl-DM4, the total plasma clearance (geometric mean [CV%]) was 4.3 L/hour (63.6%)
and the geometric mean terminal phase half-life was 5.0 days.
Metabolism
The monoclonal antibody portion of mirvetuximab soravtansine-gynx is expected to be metabolized into small
peptides by catabolic pathways. Unconjugated DM4 and S-methyl-DM4 undergo metabolism by CYP3A4. In
Reference ID: 5077370
human plasma, DM4 and S-methyl DM4 were identified as the main circulating metabolites, accounting for
approximately 0.4% and 1.4% of mirvetuximab soravtansine-gynx AUCs, respectively.
Excretion
S-methyl DM4 and DM4-sulfo-SPDB-lysine were detected in urine within 24 hours of infusion as the main
metabolites.
Specific Populations
No clinically significant differences in the pharmacokinetics of mirvetuximab soravtansine-gynx were observed
based on age (34 to 89 years), body weight (36 to 136 kg), mild hepatic impairment (total bilirubin ≤ULN and
any AST >ULN or total bilirubin >1 to 1.5 times ULN and any AST), or mild to moderate renal impairment
(CLcr ≥30 and <90 mL/min).
The pharmacokinetics of ELAHERE in patients with moderate to severe hepatic impairment (total bilirubin
>1.5 ULN with any AST) or severe renal impairment (CLcr 15 to 30 mL/min) is unknown.
Drug Interaction Studies
Clinical studies and model informed approaches
No clinical studies evaluating the drug-drug interaction potential of mirvetuximab soravtansine-gynx have been
conducted.
However, in 3 clinical trials, there were no differences in exposure between patients who received concomitant
weak or moderate CYP3A4 inhibitors or P-glycoprotein (P-gp) inhibitors and those who did not.
In Vitro Studies
Cytochrome P450 (CYP) Enzymes: Unconjugated DM4 is a time-dependent inhibitor of CYP3A4.
Unconjugated DM4 and S-methyl DM4 are not direct inhibitors of CYP1A2, CYP2B6, CYP2C8, CYP2C9,
CYP2C19, CYP2D6, or CYP3A. DM4 and S-methyl DM4 are not inducers of CYP1A2, CYP2B6, or
CYP3A4.
Transporter Systems: Unconjugated DM4 and S-methyl DM4 are substrates of P-gp but are not inhibitors of P-
gp.
12.6 Immunogenicity
As with all therapeutic proteins, there is potential for immunogenicity. The detection of antibody formation
against mirvetuximab soravtansine-gynx is highly dependent on the sensitivity and specificity of the assay. The
observed incidence of anti-drug antibodies (including neutralizing antibody) is highly dependent on the
sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the
incidence of anti-drug antibodies in the studies described below with the incidence of anti-drug antibodies to
mirvetuximab soravtansine-gynx in other studies.
With a median duration of treatment of 4.3 months in Studies 0417, 0401, and 0403, a total of 55/423 (13%)
ovarian cancer patients treated with mirvetuximab soravtansine-gynx at 6 mg/kg AIBW had at least 1 post-
baseline positive sample for anti-mirvetuximab soravtansine-gynx antibodies. Of those patients, 28/423 patients
(7%) had developed treatment-emergent ADA and 3/423 patients (0.7%) had treatment-enhanced ADA.
Neutralizing antibodies were detected in 24/423 (6%) of patients.
Because of the low occurrence of anti-mirvetuximab soravtansine-gynx antibodies, the effect of these antibodies
on the pharmacokinetics, efficacy, and/or safety of mirvetuximab soravtansine-gynx is unknown.
Reference ID: 5077370
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity studies have not been conducted with mirvetuximab soravtansine-gynx or DM4.
DM4 and the metabolite, S-methyl DM4, were clastogenic in the in vivo rat bone marrow micronucleus study.
DM4 and S-methyl DM4 were not mutagenic in the bacterial reverse mutation (Ames) assay.
Fertility studies have not been conducted with mirvetuximab soravtansine-gynx or DM4.
14 CLINICAL STUDIES
The efficacy of ELAHERE was evaluated in Study 0417 (NCT04296890), a single-arm trial of patients with
FRα positive, platinum-resistant epithelial ovarian, fallopian tube, or primary peritoneal cancer (n=106).
Patients were permitted to receive up to three prior lines of systemic therapy. All patients were required to have
received prior bevacizumab. The trial enrolled patients whose tumors were positive for FRα expression as
determined by the VENTANA FOLR1 (FOLR1-2.1) RxDx Assay. Patients were excluded if they had corneal
disorders, ocular conditions requiring ongoing treatment, Grade >1 peripheral neuropathy, or noninfectious
interstitial lung disease.
Patients received ELAHERE 6 mg/kg (based on adjusted ideal body weight) as an intravenous infusion every 3
weeks until disease progression or unacceptable toxicity. Tumor response assessments occurred every 6 weeks
for the first 36 weeks and every 12 weeks thereafter.
The major efficacy outcome measures were investigator-assessed overall response rate (ORR) and duration of
response (DOR) evaluated according to Response Evaluation Criteria in Solid Tumors (RECIST), version 1.1.
The efficacy evaluable population included 104 patients with platinum-resistant disease, who had measurable
disease, and received at least one dose of ELAHERE. In these 104 patients, the median age was 62 years (range:
35 to 85); 96% were White, 2% were Asian, and 2% did not have race reported. Two percent of patients were
Hispanic or Latino. All patients had an ECOG PS of 0 (57%) or 1 (43%). Ten percent of patients had received 1
prior line of systemic therapy, 39% of patients had received 2 prior lines of systemic therapy, and 50% of
patients had received 3 prior lines of systemic therapy. All patients had received prior bevacizumab and 47%
had received a prior PARP inhibitor.
Efficacy results for Study 0417 are summarized in Table 7.
Table 7: Efficacy Results in Study 0417
ELAHERE
(N=104)
Confirmed Overall Response Rate
a
(95% CI)
31.7%
(22.9, 41.6)
Complete response rate 4.8%
Partial response rate 26.9%
Duration of Response N=33
Median duration of response, months
(95% CI)
6.9
(5.6, 9.7)
a
Investigator assessment.
Response assessment results using independent radiology review were consistent with investigator assessment.
Reference ID: 5077370

15 REFERENCES
1
“OSHA Hazardous Drugs.” OSHA. http://www.osha.gov/SLTC/hazardousdrugs/index.html
16 HOW SUPPLIED/STORAGE AND HANDLING
How Supplied
Each ELAHERE (mirvetuximab soravtansine-gynx) injection carton (NDC 72903-853-01) contains:
• One single-dose vial containing 100 mg of mirvetuximab soravtansine-gynx in 20 mL (5 mg/mL) of
clear to slightly opalescent, colorless sterile solution.
Storage and Handling
Store ELAHERE vials upright in a refrigerator at 2°C to 8°C (36°F to 46°F) until the time of preparation in the
original carton to protect from light.
Do not freeze or shake.
ELAHERE is a hazardous drug. Follow applicable special handling and disposal procedures
1
.
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Medication Guide).
Ocular Disorders
Inform patients about the need for eye exams before and during treatment with ELAHERE.
Advise patients to contact their healthcare provider promptly if they experience any visual changes. Advise
patients to use steroid eye drops and artificial tear substitutes [see Dosage and Administration (2.3) and
Warnings and Precautions (5.1)].
Pneumonitis
Advise patients to immediately report new or worsening respiratory symptoms [see Dosage and Administration
(2.3) and Warnings and Precautions (5.2)].
Embryo-Fetal Toxicity
Advise pregnant women and females of reproductive potential of the potential risk to a fetus. Advise female
patients to inform their healthcare provider of a known or suspected pregnancy [see Use in Specific Populations
(5.4, 8.1, 8.3)].
Advise females of reproductive potential to use effective contraception during treatment with ELAHERE and
for 7 months after the last dose [see Use in Specific Populations (8.1, 8.3)].
Lactation
Advise women not to breastfeed during treatment with ELAHERE and for 1 month after the last dose [see Use
in Specific Populations (8.2)].
Manufactured by:
ImmunoGen, Inc.
Waltham, MA 02451
1-781-895-0600
U.S. License XXXX
Reference ID: 5077370
ELAHERE is a trademark of ImmunoGen, Inc.
©
2022 ImmunoGen, Inc.
All rights reserved.
Reference ID: 5077370
MEDICATION GUIDE
ELAHERE (el-ah-HERE)
(mirvetuximab soravtansine-gynx)
injection, for intravenous use
What is the most important information I should know about ELAHERE?
ELAHERE can cause serious side effects, including:
• Eye problems. Eye problems are common with ELAHERE and can also be severe. Tell your healthcare provider
right away if you develop any eye problems during treatment with ELAHERE, including blurred vision, dry eyes,
sensitivity to light, eye pain, or new or worsening vision changes.
o Your healthcare provider will send you to see an eye care professional to check your eyes before you start
treatment with ELAHERE, during treatment with ELAHERE, and as needed for any worsening signs and
symptoms of eye problems.
o Your healthcare provider will prescribe steroid eye drops and lubricating eye drops before you start and during
your treatment with ELAHERE. You should use eye drops as directed by your healthcare provider.
o Do not wear contact lenses throughout your treatment with ELAHERE unless you are told to use them by your
healthcare provider.
See “What are the possible side effects of ELAHERE?” for more information about side effects.
What is ELAHERE?
ELAHERE is a prescription medicine used to treat adults with folate receptor-alpha positive ovarian cancer, fallopian tube
cancer, or primary peritoneal cancer who:
• have not responded to or are no longer responding to treatment with platinum-based chemotherapy and
• have received 1 to 3 prior types of chemotherapy.
Your healthcare provider will perform a test to make sure that ELAHERE is right for you.
It is not known if ELAHERE is safe and effective in children.
Before receiving ELAHERE, tell your healthcare provider about all of your medical conditions, including if you:
• have vision or eye problems.
• have liver problems.
• are pregnant or plan to become pregnant. ELAHERE can harm your unborn baby. Tell your healthcare provider
right away if you become pregnant or think you may be pregnant during treatment with ELAHERE.
Females who are able to become pregnant:
o Your healthcare provider should do a pregnancy test before you start treatment with ELAHERE.
o You should use an effective birth control (contraception) during treatment and for 7 months after your last dose of
ELAHERE.
• are breastfeeding or plan to breastfeed. It is not known if ELAHERE passes into your breast milk. Do not
breastfeed during treatment and for 1 month after your last dose of ELAHERE.
Tell your healthcare provider about all the medicines you take, including prescription and over-the-counter
medicines, vitamins, and herbal supplements. Taking certain other medicines during treatment with ELAHERE may
cause side effects.
How will I receive ELAHERE?
• ELAHERE will be given to you by infusion into your vein (intravenous or IV).
• Before each dose of ELAHERE you will receive medicines to help prevent infusion related reactions, nausea, and
vomiting.
• ELAHERE is usually given every 3 weeks (21-day cycle). Your healthcare provider will decide how many cycles you
need.
What are the possible side effects of ELAHERE?
ELAHERE can cause serious side effects, including:
• See “What is the most important information I should know about ELAHERE?”
• Lung problems (pneumonitis). ELAHERE can cause severe or life-threatening inflammation of the lungs that may
lead to death. Tell your healthcare provider right away if you get new or worsening symptoms, including trouble
breathing, shortness of breath, cough, or chest pain.
• Peripheral neuropathy. You may develop nerve problems called peripheral neuropathy during treatment with
ELAHERE. Your healthcare provider will monitor you for signs and symptoms of nerve problems.
Tell your healthcare
provider if you get new or worsening numbness or tingling in your hands or feet or muscle weakness.
The most common side effects of ELAHERE include:
• feeling tired
• diarrhea
• increased liver enzymes in the blood
• decreased albumin level in the blood
Reference ID: 5077370

• nausea
• constipation
• stomach-area (abdominal) pain • decreased magnesium level in the blood
• decreased red or white blood cell counts
Your healthcare provider may change your dose of ELAHERE, delay treatment, or completel
y stop treatment if you have
certain side effects.
These are not all of the possible side effects of ELAHERE.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.
General information about the safe and effective use of ELAHERE.
Medicines are sometimes prescribed for purposes other than those listed in a Medication Guide. If you would like more
information about ELAHERE, talk with your healthcare provider. You can ask your pharmacist or healthcare provider for
information about ELAHERE that is written for healthcare professionals.
What are the ingredients in ELAHERE?
Active ingredient: mirvetuximab soravtansine-gynx
Inactive ingredients: glacial acetic acid, polysorbate 20, sodium acetate, sucrose, Water for Injection.
Manufactured by: ImmunoGen, Inc., Waltham, MA 02451
U.S. License XXXX
ELAHERE
TM
is a trademark owned by ImmunoGen, Inc.
©
2022 ImmunoGen, Inc.
For more information, go to www.immunogen.com or call 1-833-486-4646.
This Medication Guide has been approved by the U.S. Food and Drug Administration. Issued 11 2022
Reference ID: 5077370
